Abstract
Moringa oleifera is a tree species of Asian origin distributed in various countries around the world. It is considered a multipurpose plant, and its leaves are widely used by the population as food and herbal medicine. However, there are still gaps in the information regarding the phytochemical compounds of the species' leaves. Thus, the aim of this study was to perform the anatomical and histochemical characterization of M. oleifera leaflets. For this analysis, the leaflets were processed, and histochemical tests were conducted according to the usual methodology in Plant Anatomy. Moringa leaflets have anomocytic and actinocytic stomata with an amphistomatic organization, collateral vascular bundles, asymmetric mesophyll, and the presence of idioblasts in the epidermis and mesophyll. The presence and histolocalization of calcium oxalate druses, total phenolic compounds, flavonoids, lipids, proteins, pectins, mucilages, and alkaloids, which are substances that may confer biological activities to the species, were evidenced. These results contribute to expanding the anatomical knowledge of the species and enable the identification of compounds for future bioprospecting studies.
Moringa oleifera; Bioactive compounds; Histochemistry; Pharmacobotany; Leaflet
Introduction
Moringa is a tree of the species Moringa oleifera Lamark, from the monogenous family Moringaceae. Other common names it holds are "tree of life" and "miracle tree," due to its ethnobotanical value1. This species is native to the western and southern regions of the Himalayas, in India and Bangladesh. Currently, it is distributed and cultivated in various countries in Africa, South America, and Asia. This species was introduced to Brazil in 1950 for urban afforestation1.
Moringa is considered a versatile plant due to the numerous uses people make of it, such as combating hypertension, diabetes, fever, and malaria, among others. Additionally, it is also used for nutrition, either in recipes or to fortify other foods2. Thus, due to the ethnobotanical applications attributed to the parts of moringa, the species gained prominence in the commercial sector, which expanded access to moringa-derived products, especially its leaves, which have a wide range of medicinal and dietary applications2-4. However, in Brazil, the commercialization and advertising of M. oleifera were prohibited by the National Health Surveillance Agency (ANVISA), due to the sale of supplements containing this plant with various therapeutic claims not allowed for food, in addition to the lack of safety evidence for the species (Resolution Nº 1478/2019)5.
The leaves represent the primary organs of interest in M. oleifera, both medicinally and nutritionally. However, scientific literature lacks comprehensive studies on the anatomy and histochemistry of these leaves, resulting in recent informational gaps6-8. Thus, considering the plasticity of plant secondary metabolism, detailed analysis of the anatomical and histochemical aspects of plant structures is essential to understand the processes of compound production and storage at the cellular and tissue levels. Studies of this nature can not only promote a deeper understanding of the anatomy and physiology of M. oleifera, but can also be crucial in guiding pharmacognostic, molecular and bioprospecting investigations related to the leaves of this species. Furthermore, it significantly contributes to the establishment of regulatory standards ensuring the quality and identity of the species9,10. Therefore, the present study aimed to provide a comprehensive anatomical description of M. oleifera leaflets while identifying the groups of bioactive compounds located within leaf tissues through histochemical techniques.
Materials and Methods
Collection of plant material
Samples were obtained from four trees of Moringa oleifera from the state of Rio de Janeiro, Brazil, under 22°45'4''S 43°26'56''W (Chavascal, Nova Iguaçu), 22°53'4"S 43°18'52"W (Quintino Bocaiúva, Rio de Janeiro), 22°53'59"S 43°06'36"W (Icaraí, Niterói) and 22°45'42.8"S 43°28'30"W (Luz, Nova Iguaçu). The leaves were collected between the third and fifth branching of the tree stem, and the leaflets were extracted between the second and third primary leaflets from the leaf. The samples were deposited in the Rio de Janeiro Botanical Garden Herbarium (RB) (registration numbers RB 800.347, RB 806.773, RB 806.774 and RB 806.778).
Anatomical and histochemical study
For anatomical and histochemical analyses, samples from fresh leaflets were cut by hand in transverse sections, with a steel blade. The histochemical tests conducted were performed according to specific protocols and publications11,12. Safrablau was used to stain the primary and secondary cell walls; 0.05% toluidine blue and 0.02% ruthenium red to detect pectins and mucilages; sudan IV to stain total lipids; 0,25% coomassie brilliant blue to stain total proteins; 10% ferric chloride and 10% potassium dichromate to detect total phenolic compounds; 5% potassium hydroxide to detect flavonoids; 0,5% hydrochloric vanillin to detect tannins; Dragendorff reagent and Wagner reagent to detect alkaloids. Additionally, a crystal solubility test was performed using 7% acetic acid and 7.15% hydrochloric acid in different histological sections, to verify the crystals chemical composition. The diaphanization technique was used to observe the epidermis, with 15% sodium hydroxide and clarification in 50% sodium hypochlorite, followed by staining with 0.5% toluidine blue.
In general, reagents used in plant anatomy can bind to more than one substance, making complementary tests. Therefore, triplicates of each histochemical test were conducted, and two distinct reagents were used to histolocalize the same compound. In the case of the protein test, as only one reagent was used, the technique was performed in sextuplicate. Unstained sections were used as a negative control. For the tests with ferric chloride, potassium dichromate and hydrochloric vanillin, clarification was not performed to prevent 30% hypochlorite from causing interference13.
Analytical observation
Histological slides were prepared using 50% glycerin. Sections were analyzed with a Nikon Eclipse E100 light microscope and the images obtained were recorded with ImageJ software. The colors observed in the histochemical tests were described using the hexadecimal color code (HEX), based in RGB (red, green, blue), in order to increase the accuracy of the staining reports. Thus, a color scale with codes corresponding to the results of each reagent was used with the help of a free color identification website14. The technique used to optimize result presentation through color coding was adapted from the study by Ribeiro and Leitão15.
Results and Discussion
M. oleifera has a compound, tripinnate, feather-shaped leaf (FIGURE 1a), containing a petiole, followed by the primary rachis, from which the primary leaflets (pinna) depart. The primary leaflet is composed of a secondary (pinnule) and tertiary leaflet (pinnulet) (FIGURE 1b). For anatomical analysis, leaflets extracted from the middle region of the leaves were selected.
The summary of anatomical and histochemical analyses is presented in TABLE 1 and the color guide used to represent the histochemical results is shown in FIGURE 2.
: Reagents used in the histochemical localization of compounds in the abaxial (aba) and adaxial (ada) epidermis, palisade (pal) and spongy (spo) parenchyma, collenchyma (Col), xylem (xyl), and phloem (phl) in the vascular bundles (VB) of M. oleifera leaflets, indicating negative result (-), positive result for isolated or grouped cells (+) and intense positive (++) for reaction in throughout the tissue.
: Hexadecimal color system (HEX) representing the colors resulting from the histochemical reagents in the leaflet of M. oleifera.
M. oleifera leaflets are dorsiventral, with collateral vascular bundles (FIGURE 3a, b). In the midrib, angular collenchyma is present (FIGURE 3a, b, c). In the mesophyll, there are one or two layers of palisade parenchyma, vascular bundles and idioblasts containing druses (FIGURE 3d, e). The tests showed that such druses are soluble in hydrochloric acid and insoluble in acetic acid, thus being composed of calcium oxalate.
The epidermis is uniseriate, with juxtaposed, polygonal cells, where the abaxial cells are generally smaller than the adaxial ones (FIGURE 3b, d). The adaxial epidermis cells are rectangular or round shaped, while the abaxial epidermis cells have an ovoid and compact shape (FIGURE 3b, d). Unicellular tector trichomes with a rounded apex are sparsely present (FIGURE 3f).
Paradermic sections show sinuous anticlinal walls in the epidermis, especially on the abaxial face (FIGURE 3g, h). Moringa leaflets are amphistomatic, with anomocytic and actinocytic stomata, which are more frequent on the abaxial surface (FIGURE 3g). In other studies, amphistomatic leaflets were also reported6,16, while in another, only hypostomatic leaflets were observed7. Below the adaxial epidermis, there are secretory cavities that expand towards the mesophyll (FIGURE 3b). In a paradermal section, such structures can be observed through transparency (FIGURE 3h). In other study6, the presence of secretory structures was also observed in the adaxial epidermis, stained with toluidine blue, indicating mucilage. Such differences can be explained by the fact that the trees were cultivated in different locations. Geographic variations are capable of structurally influencing plant species17,18.
Secretory idioblasts are present in the parenchyma, evidenced by safrablau (FIGURE 3b, c) and by toluidine blue (FIGURE 4a, b) in cells of the spongy parenchyma, being more frequent close to the midrib. It is noteworthy that although safrablau is a dye used to highlight cell walls, it is also capable of binding to non-structural phenolic substances19. As for toluidine blue, it highlights pectins and mucilages through metachromasia and reacts differently depending on the tissue composition15.
: Cross sections of the M. oleifera leaflet showing results from Ruthenium Red, Toluidine Blue, Suvan IV and Coomassie Blue reagents.
The presence of pectins was evidenced by ruthenium red in all leaflet cells, since these substances are components of the cell wall (FIGURE 4c-e). However, there was more intense marking in secretory idioblasts present in the midrib, spongy parenchyma and secretory cavities, which indicated the presence of mucilage in these structures (FIGURE 4c-e). Mucilages are complex heteropolysaccharides, composed of long-chain carbohydrates, uronic acids and other substances and that form gels when absorbing water. These substances are soluble fibers and may have several functions in plants, such as water retention, evapotranspiration reduction, organ structure protection and carbohydrate reserve20. For humans, mucilage and pectin, which is another soluble fiber, are important for gastrointestinal function20. However, there is a paucity of studies examining the biotechnological potential of leaflet mucilage.
The presence of a cuticle in the abaxial epidermis of the midrib was evidenced by Sudan IV (FIGURE 4f). The cuticle is a lipophilic layer that acts as a barrier for water, gas and solutes diffusion, helping the plant to survive in dry environments, as well as helping to protect against pathogens21. Additionally, Sudan IV marked lipid reserves throughout the chlorenchyma (FIGURE 4g). These oily protoplasts or oleosomes may function as energy reserves for moringa22. Physicochemical analyses revealed that M. oleifera exhibits a higher content of fatty acids compared to M. peregrina23, which could be attributed to genetic as well as environmental differences between them.
Proteins were detected through Coomassie Blue staining, with higher intensity in the chlorenchyma and lower intensity in the epidermis, collenchyma, xylem, and phloem (FIGURE 4h, i). One of the reasons for the international interest in moringa as a food source lies in the high protein content of its leaves, which, according to assays, can vary from 23-28 g/100 g and encompass all essential amino acids24.
Phenolic compounds were observed in idioblasts in the epidermis, in the chlorenchyma and the collenchyma, evidenced by potassium dichromate (FIGURE 5a, b) and ferric chloride reagents (FIGURE 5c, d). Potassium hydroxide contributed to the analysis, identifying the presence of flavonoids mainly in idioblasts close to the midrib (FIGURE 5e, f). Flavonoids are polyphenolic bioactives, synthesized mainly in the vacuole, that have antioxidant activity, which is why they are targets of pharmaceutical and nutritional studies, in addition to their biological and evolutionary importance on the survival of plants under stress conditions25.
: Cross sections of the M. oleifera leaflet showing results from potassium dichromate, ferric chloride, potassium hydroxide, dragendorff and Wagner reagents.
Thus, the histochemical tests showed that M. oleifera leaflet presents secretory structure located mainly in spongy parenchyma and in the adaxial epidermis, containing mucilaginous and phenolic compounds (FIGURES 3b, c; 4a-e; 5a, c-f, h). In the study conducted Barros et al.26, it is stated that mucilage transported to the vacuole can accumulate in this compartment, displacing the cytoplasm to the cell periphery, forming a vacuole rich in mucilages. This could be the mechanism employed by mucilage cells in the leaflet of M. oleifera. Similar results were observed in other individuals of M. oleifera and in the species M. stenopetala, where they exhibited mucilaginous idioblasts in the parenchyma and around the central vein6,8.
The presence of tannins was not observed in the tests carried out with hydrochloric vanillin. The presence of alkaloids was evidenced by Dragendorff and Wagner reagents, which marked the chlorenchyma and the xylem (FIGURE 5g, h). In plants, alkaloids may be important for defense against biotic and abiotic stresses. For humans, alkaloids are interesting due to the variety of physiological effects they can have27. The study by Xie and colleagues28 reported the potential of leaf alkaloids to influence the in vitro inhibition of tumor cells.
Crystalliferous idioblasts were observed in moringa leaflets (FIGURES 3a, e; 4a, b, f-h; 5b, e), with druses frequently close to the vascular bundles. This fact is possibly related to preventing the calcium transported by the xylem from accumulating in the tissues, so that it precipitates with oxalate inside the idioblasts29. For human health, oxalate is considered an antinutritional factor because it can bind to ions, preventing their absorption by the intestinal tract. Furthermore, excessive intake of dietary oxalate is related to secondary hyperoxaluria and other renal injury conditions, which indicates caution when used in the diet of renal patients30.
Secretory structures exhibited diverse compositions of phenolic compounds, alkaloids, lipids, and mucilages. According to the literature, mucilages are known to be complex substances capable of binding to other biomolecules, as revealed throughout the results, with different chemical groups coexisting within the same cellular compartments. Such findings could contribute to directing more specific studies related to the targeted extraction of these compounds, or even to studies aimed at optimizing the production of secondary metabolites in these structures9
Conclusions
The anatomical and histochemical analysis of M. oleifera leaflets describes structural features that are applied for the species identification, such as the presence of anomocytic and actinocytic stomata, trichomes, secretory cavities below the adaxial epidermis containing mucilage and idioblasts in the mesophyll containing mucilage and phenolic compounds. Idioblasts containing druses were also observed in the mesophyll and midrib. Other substances, including lipids, proteins, pectins and alkaloids were also histolocalized in the leaflet, contributing to histochemical, metabolic, and ecological characterization of the species. The identification of the most prevalent chemical compounds in the leaflets are of great importance for future bioprospecting studies on this species, particularly due to its potential phytochemical and nutritional value.
Acknowledgments
The authors express their gratitude to the Pro-Rectorate of Extension at the Federal Institute of Education, Science, and Technology of Rio de Janeiro (IFRJ) for the scholarship granted through the Institutional Program for Initiation Scholarships in Extension (PIBIEX - proc. 23270.102730/2018-05), as well as for the research support received from the Foundation Carlos Chagas Filho Research Support of the State of Rio de Janeiro (FAPERJ) and the National Council for Scientific and Technological Development (CNPq).
References
- 1 Ghimire S, Subedi L, Acharya N, Gaire BP. Moringa oleifera: A Tree of Life as a Promising Medicinal Plant for Neurodegenerative Diseases. J Agric Food Chem [Internet]. 2021 Dec 8; 69(48): 14358-71. [cited 2023 Aug 23]. Available from: [ https://doi.org/10.1021/acs.jafc.1c04581 ].
» https://doi.org/10.1021/acs.jafc.1c04581 - 2 Dhakad AK, Ikram M, Sharma S, Khan S, Pandey VV, Singh A. Biological, nutritional, and therapeutic significance of Moringa oleifera Lam. Phytother Res [Internet]. 2019 Nov 1; 33(11): 2870-903. [cited 2023 Aug 23]. Available from: [ https://doi.org/10.1002/ptr.6475 ].
» https://doi.org/10.1002/ptr.6475 - 3 Biswas D , Nandy S , Mukherjee A , Pandey DK , Dey A . Moringa oleifera Lam. and derived phytochemicals as promising antiviral agents: A review. South Afric J Bot. 2020 Mar 1; 129: 272-82. [ https://doi.org/10.1016/j.sajb.2019.07.049 ].
» https://doi.org/10.1016/j.sajb.2019.07.049 - 4 Silva LMP, Inácio MRC, Silva GGC, Silva JMS, Luz JRD, Almeida MDG, et al. The First Optimization Process from Cultivation to Flavonoid-Rich Extract from Moringa oleifera Lam. Leaves in Brazil. Foods [Internet]. 2022 May 1 [cited 2024 Mar 9]; 11(10): 1452. Available from: [ https://doi.org/10.3390/foods11101452 ].
» https://doi.org/10.3390/foods11101452 - 5 Brasil. Ministério da Saúde. ANVISA. Resolução-RE nº 1.478, de 3 de junho de 2019. Diário Oficial da União, Seção 1, nº 106 Brasília: Resolução; Jun 4, 2019 p. 42-42. [ https://www.in.gov.br/web/dou/-/resolucao-re-n-1.478-de-3-de-junho-de-2019-152008784 ].
» https://www.in.gov.br/web/dou/-/resolucao-re-n-1.478-de-3-de-junho-de-2019-152008784 - 6 Ruiz AI, Mercado MI, Guantay ME, Ponessa GI. Anatomía e histoquímica foliar y caulinar de Moringa oleífera (Moringaceae). Boletín de la Sociedad Argentina de Botánica [Internet]. 2019 Sep 1; 54(3): 20-30. [cited 2023 Aug 23]. Available from: [ http://www.scielo.org.ar/scielo.php?script=sci_arttext&pid=S1851-23722019000300002&lng=es&nrm=iso&tlng=es ].
» http://www.scielo.org.ar/scielo.php?script=sci_arttext&pid=S1851-23722019000300002&lng=es&nrm=iso&tlng=es - 7 Vya MK. A Contribution on the Anatomical Characters of Moringa oleifera Lamk. and their significance. J Pharmacogn Phytochem. 2019; 8(2): 576-8. E-ISSN: 2278-4136. [ https://www.researchgate.net/publication/343188648_A_Contribution_on_the_Anatomical_Characters_of_Moringa_oleifera_Lamk_and_their_Significance ].
» https://www.researchgate.net/publication/343188648_A_Contribution_on_the_Anatomical_Characters_of_Moringa_oleifera_Lamk_and_their_Significance - 8 Eltaher A , Mousa AAA . Comparative Anatomical Studies on Some Moringa Species Growing in Egypt. J Agric Res. 2014; 21: 134-45. [ https://www.researchgate.net/publication/344154954_Comparative_Anatomical_Studies_on_Some_Moringa_Species_Growing_in_Egypt ].
» https://www.researchgate.net/publication/344154954_Comparative_Anatomical_Studies_on_Some_Moringa_Species_Growing_in_Egypt - 9 Yadav V, Arif N, Singh VP, Guerriero G, Berni R, Shinde S, et al. Histochemical Techniques in Plant Science: More Than Meets the Eye. Plant Cell Physiol [Internet]. 2021 Dec 3; 62(10): 1509-27. [cited 2023 Aug 23]. Available from: [ https://dx.doi.org/10.1093/pcp/pcab022 ].
» https://dx.doi.org/10.1093/pcp/pcab022 - 10 Pimentel RR, Rocha JF, Arruda RCO, Somner GV. Morfoanatomia e Histoquímica de três espécies de Paullinia L. (Sapindaceae). Rev Fitos [Internet]. 2023 Dec 20; 17(4): 479-89. [cited 2024 Mar 17]. Available from: [ https://doi.org/10.32712/2446-4775.2023.1501 ].
» https://doi.org/10.32712/2446-4775.2023.1501 - 11 Ventrela MC, Almeida AL, Nery LA, Coelho VPM. Métodos Histoquímicos Aplicados às Sementes, n o 18. Viçosa: Universidade Federal de Viçosa; 2013. 1-34 p. [ https://serieconhecimento.cead.ufv.br/edicoes/metodos-histoquimicos-aplicados-as-sementes/ ].
» https://serieconhecimento.cead.ufv.br/edicoes/metodos-histoquimicos-aplicados-as-sementes/ - 12 Figueiredo ACS, Barroso JMG, Pedro LMG, Ascensão L. Histoquímica e Citoquímica em Plantas: Princípios e Protocolos. 1 st ed. Lisboa: Faculdade de Ciências da Universidade de Lisboa, Centro de Biotecnologia Vegetal; 2007. 1-80 p. ISBN: 978-972-9348-17-4. [ https://www.uesc.br/centros/cme/arquivos/apostila_histoquimica_lisboa_2014.pdf ].
» https://www.uesc.br/centros/cme/arquivos/apostila_histoquimica_lisboa_2014.pdf - 13 Higuchi CT. Byrsonima ssp: estudo anatômico e histoquímico foliar, atividade antimicobacteriana e citotoxicidade de extratos e seus derivados. Araraquara; 2007. 70 p. Dissertação de Mestrado. [Pós-Graduação em Ciências Farmacêuticas] - Universidade Estadual Paulista, Araraquara, SP, 2007. [ http://hdl.handle.net/11449/87977 ].
» http://hdl.handle.net/11449/87977 - 14 Wibushi . Identificador de Cores Online. 2023. [ https://www.wibushi.com/br/recursos/identificador-de-cores/ ].
» https://www.wibushi.com/br/recursos/identificador-de-cores/ - 15 Ribeiro VC, Leitão CAE. Utilisation of Toluidine blue O pH 4.0 and histochemical inferences in plant sections obtained by free-hand. Protoplasma [Internet]. 2020 May 1; 257(3): 993-1008. [cited 2023 Aug 23]. Available from: [ https://link.springer.com/article/10.1007/s00709-019-01473-0 ].
» https://link.springer.com/article/10.1007/s00709-019-01473-0 - 16 Abubakar B, MuA'zu S, Khan A, Adamu A. Variability Studies as Inferred from Leaf Morpho-Stomatal Features in Moringa oleifera Lam. From Northern Nigeria. Agrosearch [Internet]. 2013 Sep 6; 13(1): 37-48. [cited 2023 Aug 23]. Available from: [ https://doi.org/10.4314/agrosh.v13i1.4 ].
» https://doi.org/10.4314/agrosh.v13i1.4 - 17 Yadav V , Arif N , Kovác J , Singh VP , Tripathi DK , Chauhan DK , et al . Structural modifications of plant organs and tissues by metals and metalloids in the environment: A review. Pl Physiol Biochem. 2021 Feb 1; 159: 100-12. [ https://doi.org/10.1016/j.plaphy.2020.11.047 ].
» https://doi.org/10.1016/j.plaphy.2020.11.047 - 18 Liu W, Zheng L, Qi D. Variation in leaf traits at different altitudes reflects the adaptive strategy of plants to environmental changes. Ecol Evol [Internet]. 2020 Aug 1; 10(15): 8166-75. [cited 2024 Mar 9]. Available from: [ https://doi.org/10.1002/ece3.6519 ].
» https://doi.org/10.1002/ece3.6519 - 19 Gancedo NC, de Lima CB, Milaneze-Gutierre MA, de Mello JCP. Pharmacobotanical study of Croton floribundus stem bark. Rodriguésia [Internet]. 2022 Apr 1; 73: e02302020. [cited 2023 Aug 22]. Available from: [ https://doi.org/10.1590/2175-7860202273032 ].
» https://doi.org/10.1590/2175-7860202273032 - 20 Dybka-Stepien K, Otlewska A, Gózdz P, Piotrowska M. The Renaissance of Plant Mucilage in Health Promotion and Industrial Applications: A Review. Nutrients [Internet]. 2021 Sep 24; 13(10): 3354. [cited 2023 Aug 23]. Available from: [ https://doi.org/10.3390/nu13103354 ].
» https://doi.org/10.3390/nu13103354 - 21 Arya GC , Sarkar S , Manasherov E , Aharoni A , Cohen H . The Plant Cuticle: An Ancient Guardian Barrier Set Against Long-Standing Rivals. Front. Plant Sci. 2021 Jun 24; 12: 663165. [ https://doi.org/10.3389/fpls.2021.663165 ].
» https://doi.org/10.3389/fpls.2021.663165 - 22 Nikiforidis CV. Structure and functions of oleosomes (oil bodies). Adv Colloid Interface Sci [Internet]. 2019 Dec 1; 274: 102039. [cited 2024 Jan 15]. Available from: [ https://doi.org/10.1016/j.cis.2019.102039 ].
» https://doi.org/10.1016/j.cis.2019.102039 - 23 Saucedo-Pompa S , Torres-Castillo JA , Castro-López C , Rojas R , Sánchez-Alejo EJ , Ngangyo-Heya M , et al . Moringa plants: Bioactive compounds and promising applications in food products. Food Res Inter. 2018 Sep 1; 111: 438-50. [ https://doi.org/10.1016/j.foodres.2018.05.062 ].
» https://doi.org/10.1016/j.foodres.2018.05.062 - 24 Benhammouche T , Melo A , Martins Z , Faria MA , Pinho SCM , Ferreira IMLPVO , et al . Nutritional quality of protein concentrates from Moringa Oleifera leaves and in vitro digestibility. Food Chem. 2021; 348: 1-8 . [ https://doi.org/10.1016/j.foodchem.2020.128858 ].
» https://doi.org/10.1016/j.foodchem.2020.128858 - 25 Wallis CM , Galarneau ERA . Phenolic Compound Induction in Plant-Microbe and Plant-Insect Interactions: A Meta-Analysis. Front Pl Sci. 2020 Dec 15; 11: 580753. [ https://doi.org/10.3389/fpls.2020.580753 ].
» https://doi.org/10.3389/fpls.2020.580753 - 26 De Barros TC, Leite VG, Pedersoli GD, Leme FM, Marinho CR, Teixeira SP. Mucilage cells in the flower of Rosales species: reflections on morphological diversity, classification, and functions. Protoplasma [Internet]. 2023 Jul 1; 260(4): 1135-47. [cited 2023 Nov 18]. Available from: [ https://doi.org/10.1007/s00709-023-01836-8 ].
» https://doi.org/10.1007/s00709-023-01836-8 - 27 Bhambhani S, Kondhare KR, Giri AP. Diversity in Chemical Structures and Biological Properties of Plant Alkaloids. Molecules. 2021 Jun 3; 26(11): 3374. [Internet]. [cited 2023 Aug 23]. Available from: [ https://doi.org/10.3390/molecules26113374 ].
» https://doi.org/10.3390/molecules26113374 - 28 Xie J , Peng LJ , Yang MR , Jiang WW , Mao JY , Shi CY , et al . Alkaloid Extract of Moringa oleifera Lam. Exerts Antitumor Activity in Human Non-Small-Cell Lung Cancer via Modulation of the JAK2/STAT3 Signaling Pathway. Evid-based Compl Alter Medic. 2021; 2021. [ https://doi.org/10.1155/2021/5591687 ].
» https://doi.org/10.1155/2021/5591687 - 29 Franceschi VR, Nakata PA. Calcium oxalate in plants: Formation and Function. Annu Rev Plant Biol. 2005 Jun 1; 56(1): 41-71. [ https://doi.org/10.1146/annurev.arplant.56.032604.144106 ].
» https://doi.org/10.1146/annurev.arplant.56.032604.144106 - 30 Huynh NK, Nguyen DHM, Nguyen HVH. Effects of processing on oxalate contents in plant foods: A review. J Food Comp Analysis. 2022 Sep 1; 112: 104685. [ https://doi.org/10.1016/j.jfca.2022.104685 ].
» https://doi.org/10.1016/j.jfca.2022.104685
-
Financing source:
FAPERJ e CNPq.
Publication Dates
- Publication in this collection
12 Mar 2025 - Date of issue
Feb 2025
History
- Received
18 Mar 2024 - Accepted
02 Nov 2024
 Anatomical and histochemical characterization of Moringa oleifera (Moringaceae) Leaflets: contributions to ethnopharmacobotanical knowledge
Anatomical and histochemical characterization of Moringa oleifera (Moringaceae) Leaflets: contributions to ethnopharmacobotanical knowledge
 Legend: a. Tripinnate leaf showing the petiole (p), primary rachis (r1º), primary leaflet (L1º); b. Primary leaflet showing secondary rachis (r2º) and subdivided into secondary leaflet (L2º), which branches into tertiary leaflet (L3º). * = region from which the leaflets were extracted for analysis. Bar: a,b: 5 cm.
Legend: a. Tripinnate leaf showing the petiole (p), primary rachis (r1º), primary leaflet (L1º); b. Primary leaflet showing secondary rachis (r2º) and subdivided into secondary leaflet (L2º), which branches into tertiary leaflet (L3º). * = region from which the leaflets were extracted for analysis. Bar: a,b: 5 cm.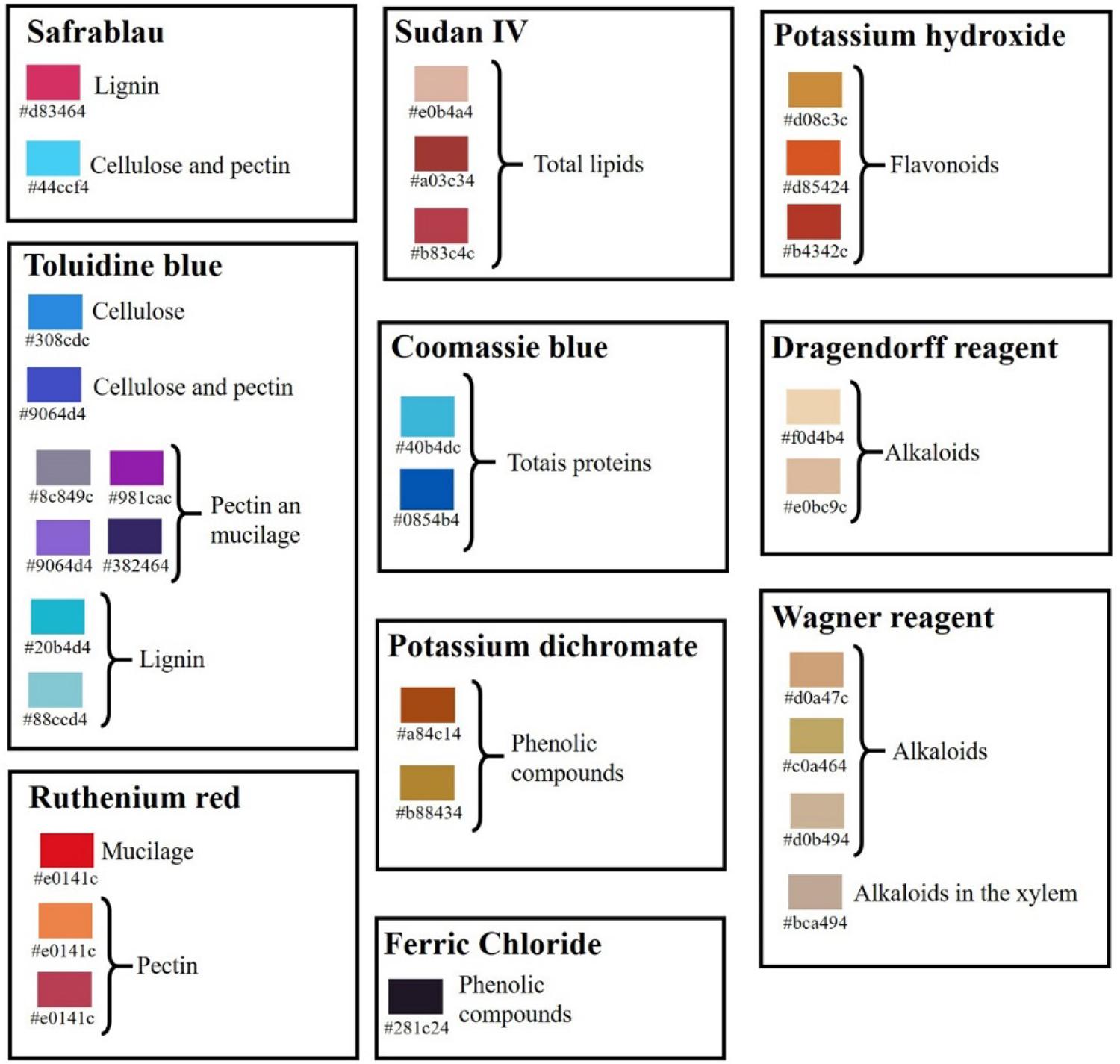 Legend: Each color is represented by a code of six numbers and letters preceded by "#", where the first two digits represent the intensity of red, the two middle digits represent the intensity of green and the last two represent the intensity of blue (#rrggbb). Histochemical results refer to safrablau, toluidine blue, ruthenium red, sudan IV, coomassie blue, potassium dichromate, ferric chloride, potassium hydroxide, Dragendorff reagent and Wagner reagent.
Legend: Each color is represented by a code of six numbers and letters preceded by "#", where the first two digits represent the intensity of red, the two middle digits represent the intensity of green and the last two represent the intensity of blue (#rrggbb). Histochemical results refer to safrablau, toluidine blue, ruthenium red, sudan IV, coomassie blue, potassium dichromate, ferric chloride, potassium hydroxide, Dragendorff reagent and Wagner reagent.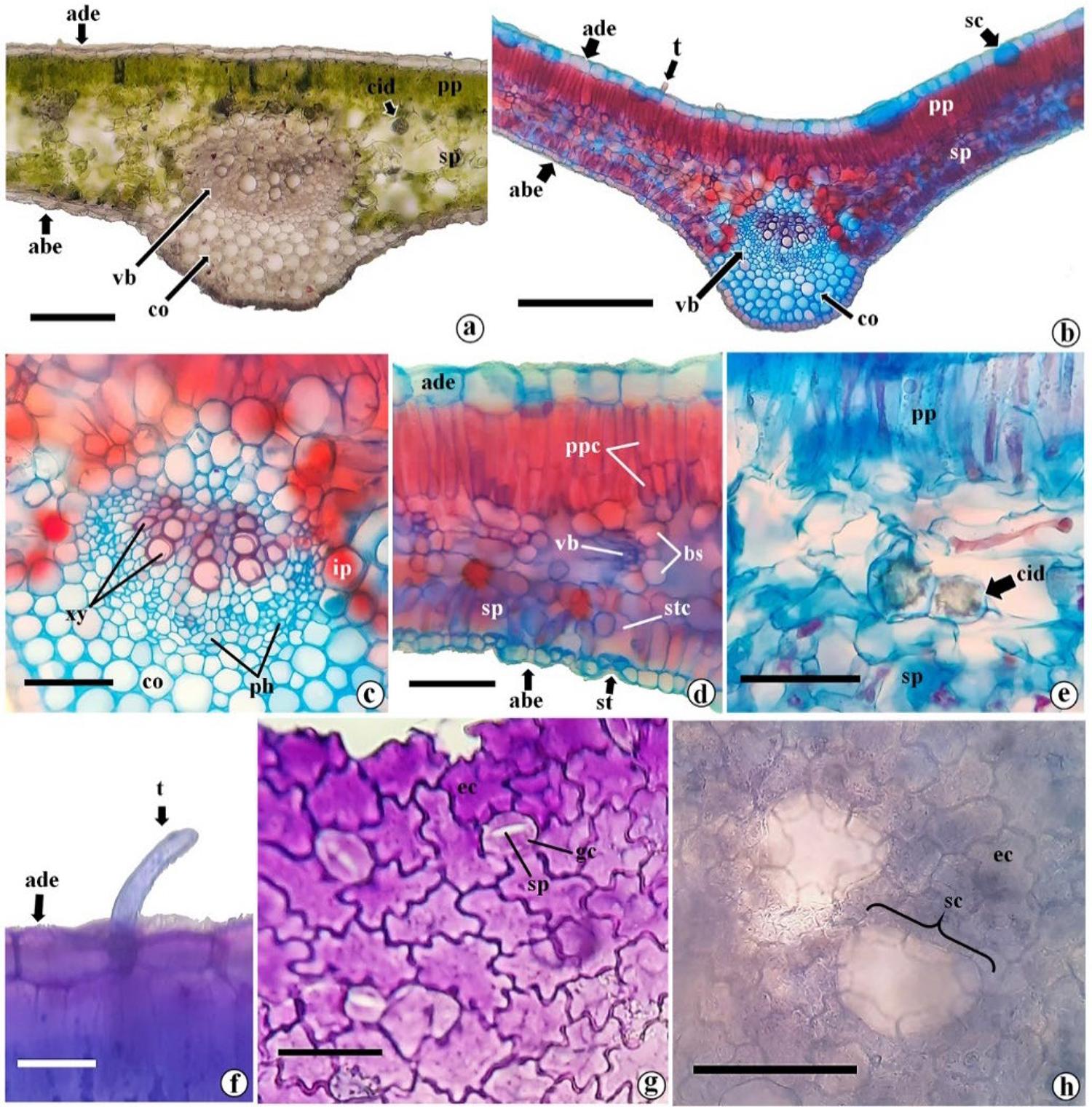 Legend: a-f. cross sections showing adaxial epidermis (ade), abaxial epidermis (abe), palisade parenchyma (pp), spongy parenchyma (sp), vascular bundle (vb), collenchyma (co); a. overview of the mesophyll without reagent (negative control); b. overview of the mesophyll with safrablau showing secretory cavities (sc) in the adaxial epidermis; c. midrib details with safrablau highlighting the xylem (xy), phloem (ph) and idioblast with phenolics (ip); d. mesophyll detail with safrablau highlighting the palisade parenchyma cells (ppc), stomata (st) together with the substomatic chamber (stc), vascular bundle with bundle sheath (bs) around it; e. detail of the mesophyll highlighting a crystalline idioblast (cid) containing drusen in the spongy parenchyma; f. cross-section of the leaflet highlighting the tector trichome (t). g, h. diaphanized paradermal sections showing epidermal cells (ec); g. paradermal section of the abaxial face of the leaflet showing the structure of the stomata, showing the guard cells, the stomatal pore (sp); h. paradermal section of the adaxial face highlighting secretory cavities below the epidermal cells. Bar: a: 100μm; b: 200μm; c-e: 50 µm; f-h: 25 µm.
Legend: a-f. cross sections showing adaxial epidermis (ade), abaxial epidermis (abe), palisade parenchyma (pp), spongy parenchyma (sp), vascular bundle (vb), collenchyma (co); a. overview of the mesophyll without reagent (negative control); b. overview of the mesophyll with safrablau showing secretory cavities (sc) in the adaxial epidermis; c. midrib details with safrablau highlighting the xylem (xy), phloem (ph) and idioblast with phenolics (ip); d. mesophyll detail with safrablau highlighting the palisade parenchyma cells (ppc), stomata (st) together with the substomatic chamber (stc), vascular bundle with bundle sheath (bs) around it; e. detail of the mesophyll highlighting a crystalline idioblast (cid) containing drusen in the spongy parenchyma; f. cross-section of the leaflet highlighting the tector trichome (t). g, h. diaphanized paradermal sections showing epidermal cells (ec); g. paradermal section of the abaxial face of the leaflet showing the structure of the stomata, showing the guard cells, the stomatal pore (sp); h. paradermal section of the adaxial face highlighting secretory cavities below the epidermal cells. Bar: a: 100μm; b: 200μm; c-e: 50 µm; f-h: 25 µm.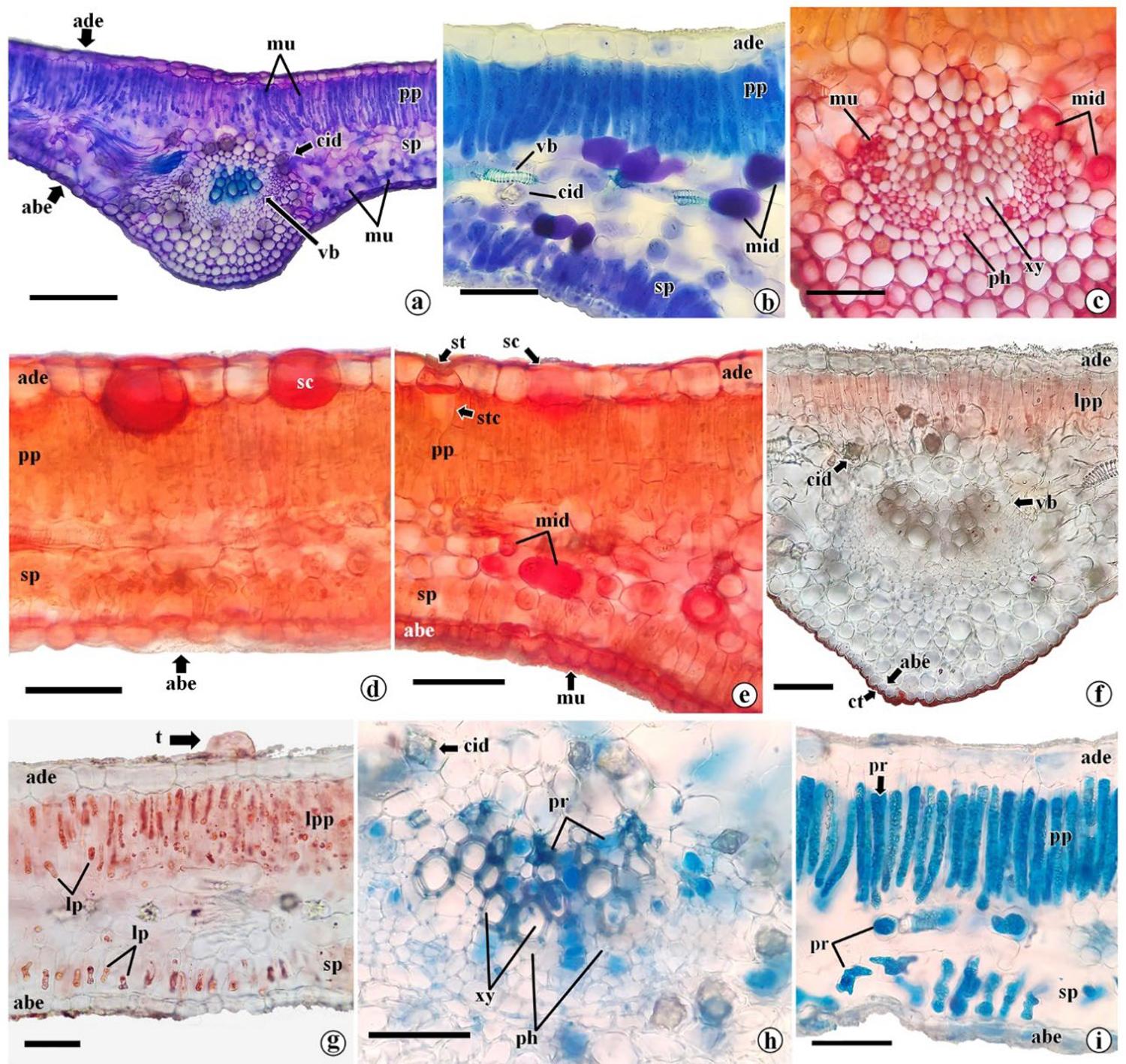 Legend: a-i. structures showing abaxial epidermis (abe), adaxial epidermis (ade), palisade parenchyma (pp), spongy parenchyma (sp), vascular bundle (vb), crystalliferous idioblast (cid); a. overview of the mesophyll with toluidine blue showing mucilage (mu) in the palisade and spongy parenchyma; b. detailed structure of chlorenchyma with toluidine blue showing vascular bundles (vb), crystalline idioblast (cid) and mucilaginous idioblast (mid); c. midrib stained with ruthenium red showing mucilage in the xylem (xy) and phloem (ph); d. detailed structure of the mesophile stained with ruthenium red showing secretory cavities (sc) with mucilage; e. chlorenchyma with ruthenium red showing mucilaginous idioblasts (mid) in the spongy parenchyma, secretory cavities (sc) with mucilage in the adaxial face, stomata (st) in the abaxial epidermis and mucilage (mu) in the abaxial epidermis; f. result of sudan IV in the midrib showing cutin (ct) and lipids in the palisade parenchyma (lpp); g. mesophyll with sudan IV highlighting lipids (lp) in palisade and spongy parenchyma cells, in addition to the presence of trichome (t); h,i. Detailed structure of midrib and mesophyll, respectively with Coomassie blue, showing proteins in xylem, phloem and parenchyma cells. stc = substomatic chamber. Bar: a: 150μm; b-i: 50 µm.
Legend: a-i. structures showing abaxial epidermis (abe), adaxial epidermis (ade), palisade parenchyma (pp), spongy parenchyma (sp), vascular bundle (vb), crystalliferous idioblast (cid); a. overview of the mesophyll with toluidine blue showing mucilage (mu) in the palisade and spongy parenchyma; b. detailed structure of chlorenchyma with toluidine blue showing vascular bundles (vb), crystalline idioblast (cid) and mucilaginous idioblast (mid); c. midrib stained with ruthenium red showing mucilage in the xylem (xy) and phloem (ph); d. detailed structure of the mesophile stained with ruthenium red showing secretory cavities (sc) with mucilage; e. chlorenchyma with ruthenium red showing mucilaginous idioblasts (mid) in the spongy parenchyma, secretory cavities (sc) with mucilage in the adaxial face, stomata (st) in the abaxial epidermis and mucilage (mu) in the abaxial epidermis; f. result of sudan IV in the midrib showing cutin (ct) and lipids in the palisade parenchyma (lpp); g. mesophyll with sudan IV highlighting lipids (lp) in palisade and spongy parenchyma cells, in addition to the presence of trichome (t); h,i. Detailed structure of midrib and mesophyll, respectively with Coomassie blue, showing proteins in xylem, phloem and parenchyma cells. stc = substomatic chamber. Bar: a: 150μm; b-i: 50 µm. Legend: a-h. structures showing abaxial epidermis (abe), adaxial epidermis (ade), palisade parenchyma (pp), spongy parenchyma (sp); a, b. mesophyll with potassium dichromate labeling idioblasts with phenolics (ip) in the epidermis, and phenolic compounds (pc) in the palisade and spongy parenchyma; c,d. mesophyll with ferric chloride labeling phenolic idioblasts and phenolic compounds in cells of palisade parenchyma, spongy parenchyma and collenchyma (co); e,f. potassium hydroxide labeling phenolic idioblasts; g. dragendorff reagent showing alkaloids (ak) in palisade and spongy parenchyma; h. Wagner reagent labeling alkaloids in parenchyma palisade, spongy parenchyma and xylem (xy). ph = phloem. Bar: a-d,f-h: 50μ; e: 300 µm.
Legend: a-h. structures showing abaxial epidermis (abe), adaxial epidermis (ade), palisade parenchyma (pp), spongy parenchyma (sp); a, b. mesophyll with potassium dichromate labeling idioblasts with phenolics (ip) in the epidermis, and phenolic compounds (pc) in the palisade and spongy parenchyma; c,d. mesophyll with ferric chloride labeling phenolic idioblasts and phenolic compounds in cells of palisade parenchyma, spongy parenchyma and collenchyma (co); e,f. potassium hydroxide labeling phenolic idioblasts; g. dragendorff reagent showing alkaloids (ak) in palisade and spongy parenchyma; h. Wagner reagent labeling alkaloids in parenchyma palisade, spongy parenchyma and xylem (xy). ph = phloem. Bar: a-d,f-h: 50μ; e: 300 µm.