Abstract
This study investigated the effect of Bauhinia forficata subsp. pruinosa leaf extract on four mycobacterial species and their biofilms, as well as its phenolic content. Phytochemical assays (total phenolic and flavonoid content, chromatographic analysis) and microbiological evaluations (minimum inhibitory concentration – MIC; minimum bactericidal concentration – MBC, time-kill curve, checkerboard assay, and antibiofilm assessment) were performed. Rutin was identified as the major compound. The extract exhibited an MIC of 0.31 mg/mL-1 for all tested strains; however, Mycobacteroides massiliense showed lower susceptibility, considering its MBC value and behavior in the time-kill curve. Additionally, the extract inhibited and eradicated biofilms of M. massiliense and Mycolicibacterium fortuitum and eradicated those of Mycobacteroides abscessus and Mycolicibacterium smegmatis when applied at the MIC value. Furthermore, the extract displayed an additive effect against M. abscessus and M. smegmatis in combination with clarithromycin and against M. massiliense when combined with imipenem. This study is the first to report the in vitro antimycobacterial and antibiofilm activity of B. forficata subsp. pruinosa leaf extract. The positive effects observed are partially attributed to the flavonol rutin. Our findings suggest its potential use as an adjuvant in the treatment of mycobacterial infections, addressing antibiotic resistance issues.
Keywords Nontuberculous mycobacteria; biofilms; drug resistance bacteria; phytochemicals; Bauhinia forficata
Introduction
Rapidly growing mycobacteria (RGM) belong to the group of non-tuberculous mycobacteria (NTM) and can cause pulmonary, skin, and soft tissue infections, as well as septicemia. Species associated with human disease are classified within the genera Mycolicibacterium (Fortuitum-Vaccae clade) and Mycobacteroides (Abscessus-Chelonae clade). These bacteria are widely distributed in the environment and have been detected in water systems and hospital equipment[1].
The prevalence of RGM infections is often underreported in tuberculosis-endemic countries such as Brazil. Treatment is typically prolonged, and despite the use of multidrug regimens, high levels of resistance have been reported. A key pathogenic feature of this group is its ability to form biofilms—aggregates of microbial cells embedded in a self-produced extracellular matrix—that adhere to surfaces and exhibit resistance to high antibiotic concentrations and standard decontamination procedures[2,3].
Plant-derived compounds have been investigated for their potential to modulate antibiotic resistance and serve as adjuvants in the treatment of infections caused by multidrug-resistant microorganisms. Brazil harbors extensive biodiversity, offering a vast reservoir of bioactive natural compounds and driving interest in the discovery of new molecules for biomedical applications. The use of medicinal plants is widespread among the Brazilian population, and the Fabaceae family is particularly noteworthy for its pharmacological potential[4].
One such species is Bauhinia forficata subsp. pruinosa (BFP), commonly known as "cow’s foot" due to the shape of its leaves. This semi-deciduous tree, native to South America, can grow up to 9 meters in height. Bioactive molecules derived from their secondary metabolism have been associated with various biological activities. Phenolic compounds, characterized by hydroxyl groups attached to aromatic rings, are abundant in plants. Among them, flavonoids exhibit remarkable structural diversity and constitute the predominant class found in different Bauhinia species, including BFP[5,6].
Aqueous and alcoholic extracts of B. forficata leaves have been traditionally used in folk medicine, and pharmacological studies have investigated their properties. Although B. forficata is widely recognized for its antidiabetic effects[7], it has also been reported to enhance Staphylococcus aureus susceptibility to norfloxacin[8] and to exhibit antibiofilm activity[9]. However, to the best of our knowledge, no studies have assessed the effects of BFP, in its traditional form of use, against RGM and their biofilms. Therefore, this study aimed to evaluate the antimycobacterial and antibiofilm activities of Bauhinia forficata subsp. pruinosa leaf extract, as well as its phenolic composition.
Material and Methods
Plant material and extract production
Bauhinia forficata subsp. pruinosa (Vogel) Fortunato & Wunderlin was collected at the Federal University of Santa Maria (UFSM), Rio Grande do Sul, Brazil (29°43'36"S, 53°43'00"W) and registered under number 9306 in the Santa Maria Herbarium (Department of Biology, UFSM). The species has been registered in the National System for the Management of Genetic Heritage and Associated Traditional Knowledge under number A09EEBA.
BFP leaves were used to prepare the extract. A total of 200 g of leaves was weighed, macerated, and immersed in 1000 mL of 70% (v/v) ethanol. The solution was stored in an amber bottle at room temperature (15–30°C) for 15 days with occasional daily stirring. After the extraction period, plant residues were discarded, and the resulting solution was filtered using number 1 filter paper (Brigitta). The filtrate was concentrated under reduced pressure at 45°C using a rotary evaporator (Fisatom 803), freeze-dried (Terroni LD1500), and stored at room temperature in a desiccator (Plenalab 03D2271.08.CC) until further use.
Determination of total phenolic compounds
Total phenolic content was determined using a modified Folin-Ciocalteu method[10]. The extract was diluted in water (50 mg/mL-1), and 1 mL of 2N Folin-Ciocalteu reagent (Exodus Scientific) was added. The mixtures were vortexed (Phoenix), incubated for 5 minutes, and then mixed with 2 mL of 20% sodium carbonate solution (Na₂CO₃, ACS Scientific). A control was prepared using only the reagents and water. Samples were incubated in the dark for an additional 10 minutes, and absorbance was measured at 730 nm using a spectrophotometer (Kasuaki IL592).
Gallic acid (Exodus Scientific) was used as the standard for constructing a calibration curve. Results were expressed as gallic acid equivalents (GAE) in mg/g of extract.
Determination of total flavonoid content
Total flavonoid content was determined using the aluminum chloride (AlCl₃) colorimetric method[10]. The extract was diluted in water (0.1 mg/mL-1) and subsequently diluted 1:3 in 70% methanol. Then, 75 μL of 5% AlCl3 solution (Dinamics) was added, and the mixture was vortexed (Phoenix) for 1 minute. The solutions were incubated in the dark for 30 minutes. A control containing only 70% methanol was prepared. Absorbance was measured at 420 nm using a spectrophotometer.
Quercetin (Sigma-Aldrich) was used as the standard for constructing a calibration curve. Results were expressed as quercetin equivalents (QE) in mg/g of extract.
Quantification of phenolic content by HPLC-DAD
Chromatographic analyses were performed using high-performance liquid chromatography with diode-array detection (HPLC-DAD)[11]. The system (Shimadzu LC-20A Nexera) included an in-line degasser (DGU-20A3), a binary pump (LC-20AB), a system controller (CBM-20A), and a PDA detector (PDA-20A, Shimadzu) with an automatic injector (SIL-20A), all controlled via LabSolutions software (Shimadzu).
Separations were performed using a Shim-Pack XR ODS II reverse-phase column (75 mm × 2.0 mm i.d., 2.1 µm particle size, Shimadzu) maintained at 35°C. The mobile phase consisted of solvent A (0.01% trifluoroacetic acid in water) and solvent B (pure methanol) in a binary gradient elution mode. The flow rate was set at 0.2 mL/min, and the injection volume was 15 µL.
Phenolic acids and flavonoids were detected at 254 nm, with identification confirmed by retention times and co-elution with authentic standards (gallic acid – Exodus Scientific; caffeic acid – Sigma-Aldrich; rutin – ACROS; quercetin – Sigma-Aldrich). Quantification was performed using the external standard method with calibration curves prepared from analytical standards in concentrations ranging from 0.05 to 100 µg/mL (R2 > 0.99). Results were expressed as mg of compound per g of dry hydroethanolic extract.
Mycobacterial strains
Four reference strains were used: Mycobacteroides abscessus (ATCC 19977), Mycobacteroides massiliense (ATCC 48898), Mycolicibacterium fortuitum (ATCC 6841), and Mycolicibacterium smegmatis (ATCC 700084). The strains were cultured on Lowenstein-Jensen agar (Himedia) at 30°C in a bacteriological incubator (Fabbe 117) until use in experiments.
Determination of minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC)
The MIC was determined using the broth microdilution method[12]. Serial twofold dilutions of BFPE in Mueller-Hinton (MH) broth (Himedia) were prepared, ranging from 4.8 to 0.03 mg/mL-1.
Bacterial inocula were standardized to 0.5 McFarland and further diluted to 5×105 CFU/mL. These were transferred to 96-well flat-bottom microplates (SPL Life Sciences) and incubated at 30°C for 72 hours. The MIC was determined as the lowest concentration inhibiting visible bacterial growth, using 1% 2,3,5-triphenyltetrazolium chloride (TTC – Vetec) as an indicator.
For MBC determination, 10 μL from wells corresponding to MIC values or higher were plated onto MH agar (Sigma-Aldrich) and incubated at 30°C for 72 hours. The lowest concentration showing no bacterial growth was considered the MBC.
Time-kill curves
Broth microdilution tests were performed, followed by plating 10 μL from each well onto MH agar at MIC, ½ MIC, and 2× MIC at 0, 24, 48, 72, and 96 hours[12]. Growth and sterility controls were included.
Plates were incubated at 30°C for 72 hours, and CFU counts were used to calculate bacterial viability. Bactericidal activity was defined as a ≥ 3-log reduction in CFU/mL compared to the untreated control.
Checkerboard assay
The interaction between BFPE and antibiotics commonly used against mycobacteria was assessed based on Nikolic et al.[13]. MICs of clarithromycin (Pharma Nostra), ciprofloxacin (Sigma-Aldrich), and imipenem (ABL Brazil) were determined.
A checkerboard microdilution assay was performed, and the Fractional Inhibitory Concentration Index (FICI) was calculated as: . FICI values were interpreted as follows: ≤0.5: Synergistic; 0.5 – 1.0: Additive; 1.0 – 2.0: Indifferent; > 2.0: Antagonistic.
Action on biofilm
The tests described below were adapted from those reported by Bonez et al.[14] and were performed in triplicate in two independent experiments.
Inhibition assay
BFPE was evaluated for its ability to inhibit biofilm formation in the four RGM species selected for this study. In 12-well polystyrene plates (IONLAB), 1 mL of MH broth containing 5×10⁵ CFU/mL of each RGM was added, along with 1 mL of BFPE at the MIC concentration. The plates were sealed with Parafilm® and incubated at 30°C for 7 days.
Disruption assay
For this assay, 1 mL of MH broth containing 5×10⁵ CFU/ mL of each RGM was added to 12-well polystyrene plates, which were sealed with Parafilm® and incubated at 30°C for 7 days. After biofilm formation, 1 mL of BFPE was added to each well at the MIC concentration. The plates were sealed again and incubated for an additional 24 hours.
Quantification
The crystal violet staining method was used to quantify biofilm biomass. After incubation with BFPE, the wells were washed with sterile Milli-Q water to remove planktonic cells. Then, 2 mL of 0.1% crystal violet solution was added, and the plates were left to stand for 10 minutes. After another washing step, 2 mL of absolute ethanol was added to each well and incubated for 15 minutes. The plates were then transferred to disposable cuvettes, and absorbance was measured at 570 nm using a spectrophotometer (Hitachi U-1800). The results were expressed as the percentage of biofilm formation relative to the positive control.
Statistical analysis
Data were expressed as mean ± standard deviation and analyzed using GraphPad Prism (version 8). Statistical significance was determined using one-way analysis of variance (ANOVA) followed by Dunnett's post hoc test or Student’s t-test. p-values < 0.05 were considered statistically significant.
Results and Discussion
Phenolic content
The yield of BFPE was 4.05% relative to the starting material. As shown in TABLE 1, BFPE contained total phenolic compounds and flavonoids. HPLC-DAD analysis identified four major bioactive compounds: gallic acid (peak 1, retention time tR = 2.85 min, 0.54 mg/g), caffeic acid (peak 2, tR = 6.32 min, 1.29 mg/g), rutin (peak 3, tR = 8.31 min, 60.96 mg/g), and quercetin (peak 4, tR = 9.45 min, 1.79 mg/g) (FIGURE 1). Among them, rutin was the most abundant metabolite, with a concentration 34 times higher than quercetin, the second most abundant compound
: Time-kill curves for (A) M. abscessus, (B) M. massiliense, (C) M. fortuitum, and (D) M. smegmatis exposed to BFPE over 0–96 hours.
Antimycobacterial effect
The MIC values were 0.31 mg/mL-1 for all tested strains. The MBC was 1.8 mg/mL-1 for M. massiliense, while for the other species, it was 1.2 mg/mL-1 The most significant findings from the bacterial time-kill curve, which assessed the relationship between concentration and exposure time to BFPE, were as follows: MIC values at 24h for M. abscessus (p < 0.0001, FIG. 1A); 2× MIC at 24h for M. massiliense (p < 0.005, FIG. 1B) and M. fortuitum (p < 0.005, FIG. 1C); and 2× MIC at 48h for M. smegmatis (p < 0.0005, FIG. 1D). The extract also exhibited additive effects against M. abscessus and M. smegmatis (in combination with clarithromycin) and M. massiliense (in combination with imipenem) (TABLE 2).
3.3. Antibiofilm activity
BFPE exhibited significant antibiofilm activity against M. massiliense and M. fortuitum (p < 0.05, FIG. 2A). The killing activity was considered excellent, as the extract acted against all tested biofilms, with particularly significant effects against M. smegmatis (p < 0.005) and M. abscessus (p < 0.0005, FIG. 2B).
This study identified phenolic metabolites and evaluated the activity of BFPE against rapidly growing mycobacteria (RGM) and their biofilms. The extract yield was considered satisfactory, as it was 4.4 times higher than the 0.92% reported by Sayago et al.[15] who also obtained a hydroethanolic extract from fresh Bauhinia forficata leaves. These authors used percolation as the extraction method, which may have contributed to the observed difference.
The phytochemical findings of this study were higher than the 69.59 mgGAE/g reported by de Miceli et al.[7] and slightly lower than the 90.1 mgGAE/g found by Franco et al.[16] in the ethyl acetate and dichloromethane fractions of B. forficata, respectively. A similar trend was observed in total flavonoid content: our results exceeded the 2.33 mgQE/g reported in the hexane fraction but were lower than the 14.9 mgQE/g found in the aqueous extract tested by the same authors. According to chromatographic analysis, the decreasing order of metabolite abundance was rutin > quercetin > caffeic acid > gallic acid, which differs from the findings of Ecker et al.[17], who analyzed the aqueous extract of B. forficata and reported a quercetin content 7.9 times higher than that of rutin.
Several factors, including climate, processing, and storage conditions, are known to influence the bioactive compound content in plants[18]. Additionally, the choice of solvent is a key factor in compound extraction, directly affecting their biological activities. In this study, ethanol was selected as the solvent to align with one of the traditional preparations of the plant, which is widely used for medicinal purposes not only in Brazil but also worldwide[19].
The presence of flavonoids in hydroethanolic extracts of Bauhinia species has been previously reported, and the synthesis of these compounds is associated with bacterial infection responses[15,20]. Rutin, a flavonol subgroup compound, was the predominant flavonoid in this study. Wang et al.[21] evaluated the antibacterial and antibiofilm activity of 10 flavonoids isolated from Klebsiella pneumoniae strains and found that rutin exhibited the greatest activity in MIC determination and biofilm inhibition.
BFPE inhibited the growth of all four Mycobacterium strains at the same MIC value in the microdilution assay, although M. massiliense exhibited lower susceptibility, as indicated by its higher MIC value and time-kill curve results. This may be attributed to the species' intrinsic resistance to antiseptics, including those used in hospital settings[22]. Furthermore, Koh et al.[23] reported that M. massiliense can develop high-level resistance through mutations in the 23S rRNA (rrl) gene.
The extract significantly inhibited and disrupted biofilms formed by M. massiliense and M. fortuitum. Considering the higher tolerance of M. massiliense to BFPE, its observed antibiofilm activity is particularly noteworthy. Additionally, M. fortuitum is considered an emerging pathogen, with infection cases becoming increasingly prevalent[24]. The extract also exhibited potent bactericidal activity against M. abscessus and M. smegmatis. Since M. abscessus infections have particularly poor treatment outcomes due to the bacterium’s ability to survive exposure to multiple antibiotic classes[25], he observed effect of BFPE is promising. Moreover, M. smegmatis biofilms have been reported to exhibit resistance to isoniazid [26], further highlighting the significance of our findings.
Biofilm formation is a critical factor that must be considered when investigating new antibacterial compounds, as bacteria within biofilms can exhibit drug resistance levels 10- to 100-fold higher than planktonic cells[27]. Flavonoids are widely distributed in plants and are less likely to induce resistance compared to conventional antibiotics[21]. Rutin has been shown to inhibit quorum sensing genes, a bacterial communication system that regulates a range of physiological activities within biofilms and plays a crucial role in bacterial survival[28].
However, it is important to note that rutin alone does not fully account for the antibiofilm activity of BFPE. The therapeutic effects of medicinal plants often result from the synergistic interactions of multiple constituents. The potential of BFPE as an adjunct therapy is further supported by its compatibility with clarithromycin, ciprofloxacin, and imipenem, as no antagonistic effects were observed in any of the combination tests. Additionally, the extract exhibited an additive effect in two drug combinations against three mycobacterial species. Harada et al.[29] reported that clinical isolates of M. massiliense exhibit relatively higher resistance to imipenem compared to M. abscessus. In this study, BFPE reduced the imipenem concentration required to inhibit M. massiliense growth by half. Natural products that can replace or reduce the required antibiotic dose are always considered a valuable therapeutic approach, particularly in the management of RGM infections[30]. Our findings suggest that BFPE holds promise as a candidate for combination therapy, potentially contributing to improved treatment strategies against mycobacterial infections.
Conclusion
Therefore, this study is the first to report the in vitro antimycobacterial and antibiofilm activities of the Bauhinia forficata subsp. pruinosa leaf extract. Rutin, the most abundant flavonol identified, is likely to contribute, at least in part, to the observed bioactivity. BFPE demonstrated efficacy against RGM and their biofilms while also enhancing the effectiveness of clinically relevant antibiotics by reducing their required dosage. These findings highlight its potential as an adjuvant in the treatment of mycobacterial infections, offering a promising strategy to mitigate bacterial resistance. Further studies, including detailed phytochemical analyses, are needed to elucidate the contribution of bioactive compounds and optimize their therapeutic applicability.
References
- 1 Gupta RS, Lo B, Son J. Phylogenomics and Comparative Genomic Studies Robustly Support Division of the Genus Mycobacterium into an Emended Genus Mycobacterium and Four Novel Genera. Front Microbiol. 2018; 13(9): 67. Available at: [ https://doi.org/10.3389/fmicb.2018.00067 ].
» https://doi.org/10.3389/fmicb.2018.00067 - 2 Koo H, Falsetta ML, Klein MI. The exopolysaccharide matrix: a virulence determinant of cariogenic biofilm. J Dent Res. 2013; 92(12): 1065-73. Available at: [ https://doi.org/10.1177/0022034513504218 ].
» https://doi.org/10.1177/0022034513504218 - 3 Barai L, Saha MR, Rahman T, Sukanya M, Ferdous J, Khanduker A, et al. Pattern of rapidly growing mycobacteria (RGM) species isolated from clinical samples: A 10-year retrospective study in a tertiary care hospital of Bangladesh. Indian J Med Microbiol. 2024; 5(53): 100756. Available at: [ https://doi.org/10.1016/j.ijmmb.2024.100756 ].
» https://doi.org/10.1016/j.ijmmb.2024.100756 - 4 Ahmad F, Anwar F, Hira S. Review on medicinal importance of Fabaceae family. PhOL. 2016; 3: 151-6. Available at: [ https://pharmacologyonline.silae.it/files/archives/2016/vol3/PhOL_2016_3_A023_47_Ahmad.pdf ].
» https://pharmacologyonline.silae.it/files/archives/2016/vol3/PhOL_2016_3_A023_47_Ahmad.pdf - 5 Jung EP, de Freitas BP, Kunigami CN, Moreira DL, de Figueiredo NG, Ribeiro LO, et al. Bauhinia forficata Link Infusions: Chemical and Bioactivity of Volatile and Non-Volatile Fractions. Molecules. 2022; 27(17): 5415. Available at: [ https://doi.org/10.3390/molecules27175415 ].
» https://doi.org/10.3390/molecules27175415 - 6 Lorenzi H, Matos FJA. Medicinal plants in Brazil: native and exotic. 3 th ed. Nova Odessa: Plantarum Botanical Garden; 2021.
- 7 Miceli N, Buongiorno LP, Celi MG, Cacciola F, Dugo P, Donato P, et al. Role of the flavonoid-rich fraction in the antioxidant and cytotoxic activities of Bauhinia forficata Link. (Fabaceae) leaves extract. Nat Prod Res. 2016; 30(11): 1229-39. Available at: [ https://doi.org/10.1080/14786419.2015.1050671 ].
» https://doi.org/10.1080/14786419.2015.1050671 - 8 De Sousa JN, de Oliveira ABM, Ferreira AK, Silva E, de Sousa LMS, França Rocha MC, et al. Modulation of the resistance to norfloxacin in Staphylococcus aureus by Bauhinia forficata link. Nat Prod Res. 2021; 35(4): 681-5. Available at: [ https://doi.org/10.1080/14786419.2019.1590714 ].
» https://doi.org/10.1080/14786419.2019.1590714 - 9 Ferreira-Filho JCC, Marre ATO, De Sá Almeida JS, Lobo LA, Farah A, Romanos MTV, et al. Therapeutic Potential of Bauhinia forficata Link in Dental Biofilm Treatment. J Med Food. 2020; 23(9): 998-1005. Available at: [ https://doi.org/10.1089/JMF.2019.0277 ].
» https://doi.org/10.1089/JMF.2019.0277 - 10 Silva FLL , Scotti AS , Garcia ALH , Lemes MLB , Grivicich I , Reis GM , et al . Toxicological potential of Aloysia gratissima: Insights from chemical analysis and in vitro studies. J Ethnopharmacol. 2023;314: 116614. Available at: [ https://doi.org/10.1016/J.JEP.2023.116614 ].
» https://doi.org/10.1016/J.JEP.2023.116614 - 11 Menezes APS , Silva J , Fisher C , Silva FR , Reyes JM , Picada JN , et al . Chemical and toxicological effects of medicinal Baccharis trimera extract from coal burning area. Chemosphere. 2016; 146: 396-404. Available at: [ https://doi.org/10.1016/J.CHEMOSPHERE.2015.12.028 ].
» https://doi.org/10.1016/J.CHEMOSPHERE.2015.12.028 - 12 Rossi GG , Guterres KB , Moreira KS , Burgo TAL , de Campos MMA , Iglesias BA . Photo-damage promoted by tetra-cationic palladium (II) porphyrins in rapidly growing mycobacteria. Photodiagnosis Photodyn Ther. 2021; 36: 102514. Available at: [ https://doi.org/10.1016/J.PDPDT.2021.102514 ].
» https://doi.org/10.1016/J.PDPDT.2021.102514 - 13 Nikolic I, Vukovic D, Gavric D, Cvetanovic J, Aleksic Sabo V, Gostimirovic S, et al. An Optimized Checkerboard Method for Phage-Antibiotic Synergy Detection. Viruses. 2022; 14(7): 1542. Available at: [ https://doi.org/10.3390/V14071542/S1 ].
» https://doi.org/10.3390/V14071542/S1 - 14 Bonez PC , Agertt VA , Rossi GG , Siqueira FS , Siqueira JD , Marques LL , et al . Sulfonamides complexed with metals as mycobacterial biofilms inhibitors. J Clin Tuberc Other Mycobact Dis. 2021; 23: 100217. Available at: [ https://doi.org/10.1016/J.JCTUBE.2021.100217 ].
» https://doi.org/10.1016/J.JCTUBE.2021.100217 - 15 Sayago C, Camargo V, Barbosa F, Gularte C, Pereira G, Miotto S, et al. Chemical composition and in vitro antioxidant activity of hydro-ethanolic extracts from Bauhinia forficata subsp. pruinosa and B. variegata. Acta Biol Hung. 2013; 64(1): 21-33. Available at: [ https://doi.org/10.1556/ABIOL.64.2013.1.3 ].
» https://doi.org/10.1556/ABIOL.64.2013.1.3 - 16 Franco RR , Alves VHM , Zabisky LFR , Justino AB , Martins MM , Saraiva AL , et al . Antidiabetic potential of Bauhinia forficata Link leaves: a non-cytotoxic source of lipase and glycoside hydrolases inhibitors and molecules with antioxidant and antiglycation properties. Biomed Pharmacother. 2020; 123: 109798. Available at: [ https://doi.org/10.1016/J.BIOPHA.2019.109798 ].
» https://doi.org/10.1016/J.BIOPHA.2019.109798 - 17 Ecker A , Vieira FA , Prestes AS , Dos Santos MM , Ramos A , Ferreira RD , et al . Effect of Syzygium cumini and Bauhinia forficata aqueous-leaf extracts on oxidative and mitochondrial parameters in vitro. EXCLI J. 2015; 14: 1219-31 . Available at: [ https://doi.org/10.17179/EXCLI2015-576 ].
» https://doi.org/10.17179/EXCLI2015-576 - 18 Sotiropoulou ?SD, Flampouri E, Skotti E, Pappas C, Kintzios S, Tarantilis PA. Bioactivity and toxicity evaluation of infusions from selected Greek herbs. Food Biosci. 2020; 35: 100598. Available at: [ https://doi.org/10.1016/J.FBIO.2020.100598 ].
» https://doi.org/10.1016/J.FBIO.2020.100598 - 19 Farag MA , Sakna ST , El-Fiky NM , Shabana MM , Wessjohann LA . Phytochemical, antioxidant and antidiabetic evaluation of eight Bauhinia L. species from Egypt using UHPLC-PDA-qTOF-MS and chemometrics. Phytochemistry. 2015; 119: 41-50. Available at: [ https://doi.org/10.1016/J.PHYTOCHEM.2015.09.004 ].
» https://doi.org/10.1016/J.PHYTOCHEM.2015.09.004 - 20 Roy A , Khan A , Ahmad I , Alghamdi S , Rajab BS , Babalghith AO , et al . Flavonoids a Bioactive Compound from Medicinal Plants and Its Therapeutic Applications. Biomed Res Int. 2022; 2022: 5445291. Available at: [ https://doi.org/10.1155/2022/5445291 ].
» https://doi.org/10.1155/2022/5445291 - 21 Wang Z , Ding Z , Li Z , Ding Y , Jiang F , Liu J . Antioxidant and antibacterial study of 10 flavonoids revealed rutin as a potential antibiofilm agent in Klebsiella pneumoniae strains isolated from hospitalized patients. Microb Pathog. 2021; 159: 105121. Available at: [ https://doi.org/10.1016/J.MICPATH.2021.105121 ].
» https://doi.org/10.1016/J.MICPATH.2021.105121 - 22 Cheng A, Sheng WH, Huang YC, Sun HY, Tsai YT, Chen ML, et al. Prolonged postprocedural outbreak of Mycobacterium massiliense infections associated with ultrasound transmission gel. Clin Microbiol Infect. 2016; 22(4): 382.e1-382.e11. Available at: [ https://doi.org/10.1016/J.CMI.2015.11.021 ].
» https://doi.org/10.1016/J.CMI.2015.11.021 - 23 Koh WJ , Jeong BH , Jeon K , Kim SY , Park KU , Park HY , et al . Oral Macrolide Therapy Following Short-term Combination Antibiotic Treatment of Mycobacterium massiliense lung disease. Chest. 2016; 150(6): 1211-21. Available at: [ https://doi.org/10.1016/J.CHEST.2016.05.003 ].
» https://doi.org/10.1016/J.CHEST.2016.05.003 - 24 Donohue MJ. Epidemiological risk factors and the geographical distribution of eight Mycobacterium species. BMC Infect Dis. 2021; 21(1): 258. available at: [ https://doi.org/10.1186/S12879-021-05925-Y ].
» https://doi.org/10.1186/S12879-021-05925-Y - 25 Cowman S, Van Ingen J, Griffith DE, Loebinger MR. Non-tuberculous mycobacterial pulmonary disease. Eur Respir J. 2019; 54(1): 1900250. Available at: [ https://doi.org/10.1183/13993003.00250-2019 ].
» https://doi.org/10.1183/13993003.00250-2019 - 26 Teng R, Dick T. Isoniazid resistance of exponentially growing Mycobacterium smegmatis biofilm culture. FEMS Microbiol Lett. 2003; 227(2): 171-4. Available at: [ https://doi.org/10.1016/S0378-1097 (03)00584-6].
» https://doi.org/10.1016/S0378-1097 (03)00584-6 - 27 Shivaprasad DP, Taneja NK, Lakra A, Sachdev D. In vitro and in situ abrogation of biofilm formation in E. coli by vitamin C through ROS generation, disruption of quorum sensing and exopolysaccharide production. Food Chem. 2021; 341(1): 128171. Available at: [ https://doi.org/10.1016/J.FOODCHEM.2020.128171 ].
» https://doi.org/10.1016/J.FOODCHEM.2020.128171 - 28 Peng LY , Yuan M , Cui ZQ , Wu ZM , Yu ZJ , Song K , et al . Rutin inhibits quorum sensing, biofilm formation and virulence genes in avian pathogenic Escherichia coli. Microb Pathog. 2018; 119: 54-9 . Available at: [ https://doi.org/10.1016/J.MICPATH.2018.04.007 ].
» https://doi.org/10.1016/J.MICPATH.2018.04.007 - 29 Harada T, Akiyama Y, Kurashima A, Nagai H, Tsuyuguchi K, Fujii T, et al. Clinical and Microbiological Differences between Mycobacterium abscessus and Mycobacterium massiliense lung diseases. J Clin Microbiol. 2012; 50(11): 3556-61. Available at: [ https://doi.org/10.1128/JCM.01175-12 ].
» https://doi.org/10.1128/JCM.01175-12 - 30 Deepika MS, Thangam R, Sakthidhasan P, Arun S, Sivasubramanian S, Thirumurugan R. Combined effect of a natural flavonoid rutin from Citrus sinensis and conventional antibiotic gentamicin on Pseudomonas aeruginosa biofilm formation. Food Control. 2018; 90: 282-94. Available at: [ https://doi.org/10.1016/J.FOODCONT.2018.02.044 ].
» https://doi.org/10.1016/J.FOODCONT.2018.02.044
-
Funding:
This study was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil – Funding Code 001.
Publication Dates
- Publication in this collection
19 Jan 2026 - Date of issue
Jan 2026
History
- Received
08 Feb 2025 - Accepted
10 Sept 2025
 Antimycobacterial and antibiofilm activity of Bauhinia forficata subsp. pruinosa leaf extract
Antimycobacterial and antibiofilm activity of Bauhinia forficata subsp. pruinosa leaf extract
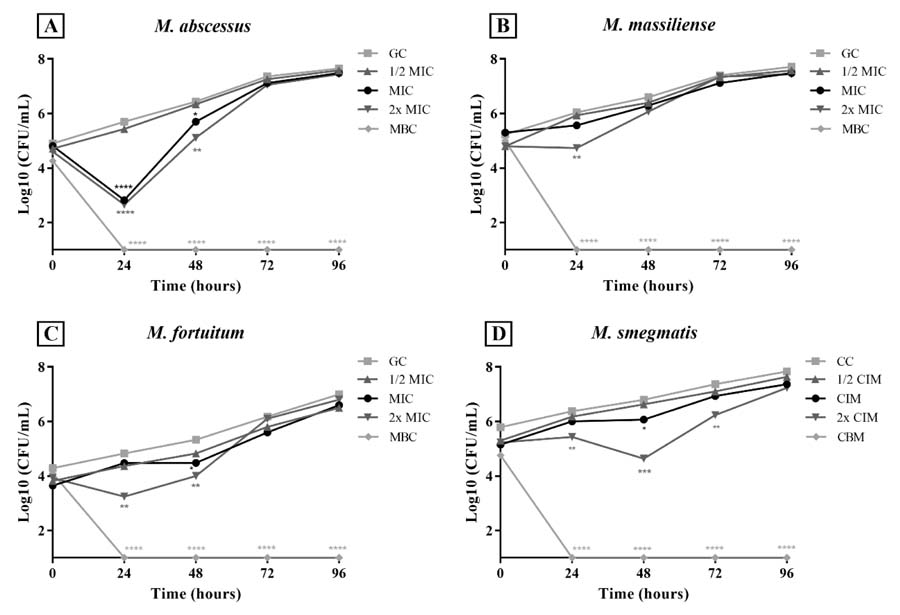 Values are expressed as a percentage of the growth control (GC) and presented as mean. MIC = minimum inhibitory concentration; 1/2 MIC = half the MIC value; 2× MIC = double the MIC value; MBC = minimum bactericidal concentration. Statistical significance was considered at p < 0.05. * p < 0.05; ** p < 0.005; *** p < 0.0005; **** p < 0.0001. Source: Authors.
Values are expressed as a percentage of the growth control (GC) and presented as mean. MIC = minimum inhibitory concentration; 1/2 MIC = half the MIC value; 2× MIC = double the MIC value; MBC = minimum bactericidal concentration. Statistical significance was considered at p < 0.05. * p < 0.05; ** p < 0.005; *** p < 0.0005; **** p < 0.0001. Source: Authors.