Abstract
The Euphorbiaceae family comprises plants with significant therapeutic potential, exemplified by the genera Dalechampia, Euphorbia, and Croton, which are recognized for their bioactive compounds and established applications in traditional medicine. Dalechampia, despite being comparatively less investigated, exhibits remarkable phytochemical diversity, encompassing diterpenes, triterpenes, flavonoids, alkaloids, and phenolic compounds. These constituents demonstrate notable antioxidant, anti-inflammatory, antimicrobial, and neuroprotective activities. This review aims to examine the pharmacological potential of Dalechampia, highlighting both its commonalities and distinctive characteristics compared with other genera in the family. While Euphorbia and Croton are extensively studied, comprehensive studies of the genus Dalechampia remain limited. Nevertheless, its isolated compounds, such as triterpenes and flavonoids, indicate promising therapeutic capabilities. Furthermore, the resin production in Dalechampia, integral to its defence mechanisms and pollinator attraction, underscores an ecological role that largely awaits thorough exploration. Comparisons with more extensively studied genera underscore the necessity for further pharmacological and toxicological investigations to elucidate the full spectrum of Dalechampia's potential applications. Future research endeavours should integrate chemical, biological, and ecological analyses to enhance the understanding of this genus, thereby contributing to the discovery of novel therapeutic agents and the sustainable management of its natural resources.
Keywords Solidago chilensis; cultivation; flavonoids; saponins
Introduction
The Euphorbiaceae family, a member of the order Malpighiales, is one of the largest families of flowering plants, encompassing approximately 16,000 species and exhibiting remarkable morphological and ecological diversity[1]. The order Malpighiales is extensive, comprising around 37 families and 716 genera[2], and accounts for approximately 40% of tree diversity in tropical forests[3]. Within this diverse order, Euphorbiaceae is notable for its complexity and wide array of life forms, including vines, herbs, shrubs, and trees, all adapted to various environments. This family has a pantropical distribution, comprising approximately 300 genera and 6,000 species globally[4]. In Brazil, specifically, 63 genera and 939 species of Euphorbiaceae are recognized[5].
First described by the French naturalist Antoine Laurent de Jussieu in 1789, the Euphorbiaceae family was initially categorized into 33 genera, with groupings based on the number of stipes[6]. Subsequently, the classification of Euphorbiaceae has undergone numerous significant revisions, notably those of Webster[7-9]. Webster proposed a modern classification system derived from morphological characteristics, which further divided the family into five subfamilies: Phyllanthoideae, Oldfieldioideae, Acalyphoideae, Crotonioideae, and Euphorbioideae.
The genus Dalechampia, formally described by Linnaeus in 1754 in honor of the French physician and botanist Jacques Dalechamp, exhibits considerable morphological diversity. It comprises approximately 130 species, with over 70 of these occurring in South America[10]. Species of Dalechampia are typically climbers or subshrubs and are characterized by distinctive inflorescences in which involucral bracts encircle both pistillate and staminate flowers.
Despite their extensive morphological diversity, research on the chemical composition and biological activity of Dalechampia species remains limited. Most studies have focused on morphological and taxonomic aspects, while investigations of bioactive compounds and their potential medicinal applications remain notably scarce. This gap underscores the critical need for more in-depth chemical studies of the genus to explore its pharmacological potential and gain a more comprehensive understanding of its ecological role.
The genus Dalechampia constitutes a significant area of inquiry for botanical, ecological, and pharmacological studies. Continued research on this genus has the potential to unveil new insights into its ecological interactions and therapeutic capabilities, thereby advancing knowledge and conserving biodiversity.
Experimental
For this review, fifty-four scientific papers focusing on the biological and chemical characterization of the genus Dalechampia were selected, all of which presented pertinent analyses, results, and conclusions. The literature search was systematically performed across five widely recognized indexed databases: Scielo, Scopus, Web of Science, PubMed, and Jstor. The inclusion criteria for the analysis were restricted to scientific publications, books, and mission reports from expeditions.
a) Search procedures
The following terms were employed for the literature search: "Dalechampia genus," "Dalechampia chemical," "Dalechampia extract," "Dalechampia metabolites," "Biological activity Dalechampia," and "phytochemistry Dalechampia." These terms were systematically combined to identify relevant associations and outcomes for characterizing the chemical composition and biological activities of Dalechampia species.
b) Inclusion and Exclusion Criteria
The inclusion criteria encompassed publications dated between 1789 and 2024, encompassing articles written in English, Portuguese, and French. Following data collection from each database, duplicate references were systematically identified and removed to prevent redundancy in the analysis. Subsequently, only studies deemed relevant to the research aim were selected, while those of insufficient methodological quality or falling outside the study's scope were excluded during the initial review process.
c) Data Analysis and Systematization
The article analysis process adhered to an integrative review methodology, commencing with an exploratory reading of the selected publications to ascertain their primary focus and scope. Subsequently, the extracted data were organized and categorized into three main sections:
-
Chemical Composition;
-
Chemical Structure of the Compounds Found; and
-
Biological Activities.
To facilitate data analysis and identify recurrent patterns, NVivo text analysis software was used to categorize the chemical substances and biological activities reported across the various Dalechampia species. This analytical approach synthesized the documented biological and chemical properties of the genus, providing a comprehensive, structured overview of current knowledge in the field.
Results and Discussion
a) Chemical composition
Species within the genus Dalechampia are characterized by the presence of specific chemical compounds, including diterpenes (1) found in their sap. These compounds are potentially toxic to animals and humans, necessitating the use of gloves during handling and the keeping of plants out of reach of children and animals[11]. Furthermore, species of the genus contain resinous compounds (2) within their inflorescences, which play a crucial role in reproduction and pollination. While Dalechampia does not produce latex, it possesses basilar glands on its stipules, which may contribute to plant defense or facilitate interactions with pollinating insects[12]. The genus also secretes triterpene-rich resins (3), which serve significant functions in both defense against herbivores and pollinator attraction. These resins, composed of mixtures of oxygenated triterpenes (4), are secreted by floral glands and serve as a reward for pollinators, such as bees, which use them for nest construction[13].
In southern Brazil, several species of Dalechampia have defensive structures that attract pollinators. D. ficifolia and D. stipulacea have urticating trichomes (5), which cause irritation on contact, as well as glandular trichomes on the involucral bracts and stipules, reinforcing their defense against herbivores. In D. stipulacea and D. stenosepala, the sepals of the pistillate flowers have glands on the distal portions, which become rigid and pointed around the fruit, warding off predators. The resinous bracteoles, common among these species, also play an important role. In D. ficifolia, the bracteoles are whitish; in D. glechomifolia and D. weddelliana, they vary between yellow-green and orange; and in D. micromeria, they are bright yellow. This resin serves as a visual attraction and reward for pollinating bees, which use it to build nests, contributing to the pollination and ecology of the species[14].
In southern Brazil, several Dalechampia species possess defensive structures that also attract pollinators. For instance, D. ficifolia and D. stipulacea exhibit urticating trichomes (5) that cause irritation upon contact, along with glandular trichomes on their involucral bracts and stipules, thereby reinforcing their defense against herbivores. In D. stipulacea and D. stenosepala, the sepals of the pistillate flowers feature glands on their distal portions, which become rigid and pointed around the fruit, effectively deterring predators. The resinous bracteoles, prevalent among these species, also fulfill a significant function. In D. ficifolia, these bracteoles are whitish; in D. glechomifolia and D. weddelliana, they range from yellow-green to orange; and in D. micromeria, they are distinctly bright yellow. This resin serves as both a visual attractant and a reward for pollinating bees, which utilize it for nest construction, consequently contributing to the pollination and ecology of the respective species[14].
The species D. stipulacea was the subject of a study conducted in 1979 in São Paulo, Brazil, by Sazima et al.[15]. This plant is characterized by two large green bracts on each inflorescence, which open only during a restricted period from 1:30 p.m. to 6:00 p.m., thereby limiting access to the flowers. A prominent gland secretes a sticky resinous compound, which is collected by two species of bees from the genus Euglossa, with Euglossa melanotricha identified as the most effective pollinator. Observations revealed that only female bees visited the flowers, while four other, less frequent bee species acted as pollen and/or resin plunderers. Additionally, assassin bugs (Apiomerus lanipes) were also observed utilizing the resin. Despite D. stipulacea's capacity for self-pollination, cross-pollination demonstrably enhances fruit production.
The study by Araújo et al.[16] investigated the chemical characterization of compounds isolated from the dried aerial parts of D. pernambucensis. The methodology employed involved extraction and fractionation using chromatographic columns with increasingly polar solvents. This approach allowed for the successful isolation of various fractions and the subsequent identification of compounds such as β-sitosterol (6), stigmasterol (in an equimolar mixture) (7), β-amyrin (8), 3-oxo-olean-9(11):12-diene (9), and 11α,12α-epoxy-3-oxo-D-friedooleanan-14-ene (10).
One of the primary compounds identified in the genus by Armbruster et al.[17] is the oxygenated triterpene resin (4), secreted by glands associated with the staminate flowers. This resin serves both as a defense mechanism against herbivores and as a reward to attract pollinating bees. This type of resin is also secreted in other vegetative parts, such as leaves and bracts, where it acts as a barrier against predators. Another defense mechanism involves glochid spines on the pistillate sepals, which protect developing seeds and fruits from predators. Additionally, trichomes containing crystals and histamine (11) are present on the leaves, providing further protection against herbivores, including insects and mammals.
The study of D. brownsbergensis by Webster and Armbruster[18] identified scattered dark-point glands on its bracts and floral structures. These glands, despite their small and inconspicuous nature, are thought to produce aromatic compounds (12) that attract male Euglossini bees, an ecological adaptation considered rare within the genus Dalechampia. These volatile compounds, characteristic of D. brownsbergensis, distinguish it from other species and signify a crucial evolutionary adaptation for the plant's specific pollination mechanism.
D. dioscoreifolia is known to contain a variety of bioactive chemical compounds, including flavonoids (13), terpenes, phenols (14), and fatty acids (15). Specifically, the plant's bracts contain resin compounds (2), while its leaves and stems contain alkaloids (16) and other phenolic compounds (14). These compounds are suggested to possess medicinal properties, such as anti-inflammatory, antioxidant, and antimicrobial activities, although further studies are required to confirm their efficacy and safety[19].
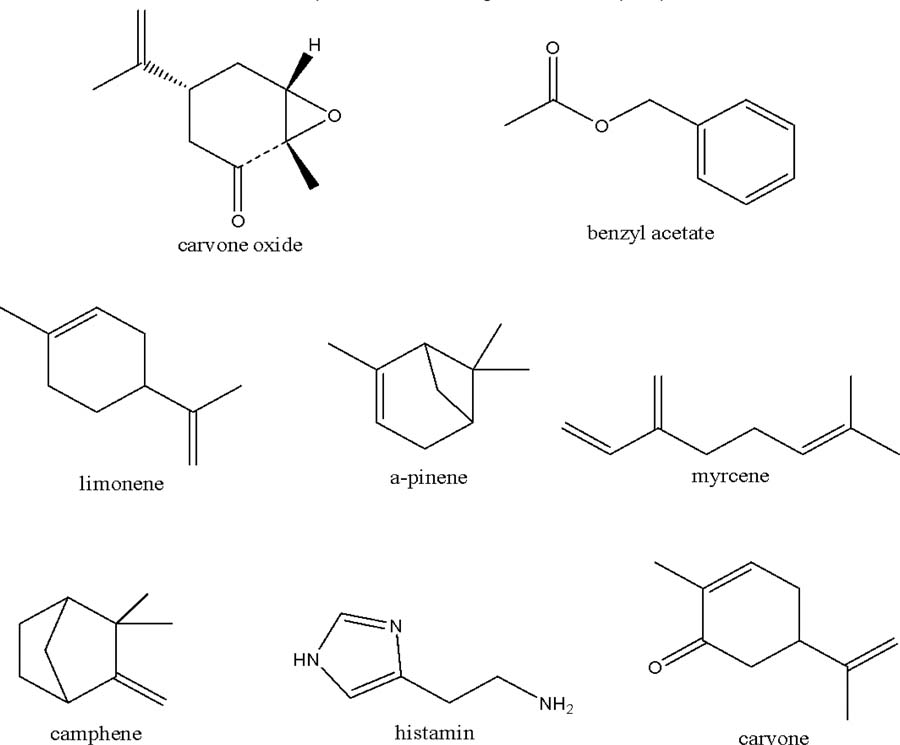
The chemical compounds identified by Armbruster et al.[20] in the fragrance collected by bees pollinating D. magnoliifolia include carvone oxide (17), benzyl acetate (18), limonene (19), α-pinene (20), myrcene (21), β-pinene/camphene (22), and carvone. Carvone oxide (17) is an aromatic compound capable of attracting pollinators, while benzyl acetate (18), noted for its sweet aroma, tends to be particularly attractive to insects. Limonene (19), a citrus-scented monoterpene, and α-pinene (20), characterized by a piney aroma, are frequently associated with the attraction of specific bee species. Myrcene (21), a terpene that enhances floral aromas, can also influence pollinator interaction. Furthermore, the compounds β-pinene/camphene (22) are integral to the aromatic mixture, potentially increasing overall attractiveness to pollinators. Finally, carvone, possessing similarly attractive properties, contributes to the complexity of the flowers' fragrance. These compounds are crucial to the interaction between D. magnoliifolia and its pollinators, as their presence and synergistic effects can affect both the frequency and effectiveness of pollination.
Srivastava et al.[21] investigated the chemical and antioxidant properties of D. scandens var. Cordofona. Their research focused on extracts derived from the plant's leaves and stem, obtained using both polar (methanol) and apolar (toluene) solvents. Phytochemical analyses revealed the presence of various bioactive compounds, including alkaloids (16), carbohydrates, proteins, phenolics (14), flavonoids (13), saponins (23), tannins (24), glycosides (25), quinones (26), steroids, terpenoids, and diterpenes (1). Notably, phenolic compounds (14) and flavonoids (13) were detected at higher concentrations in the methanolic extracts, which also demonstrated significant antioxidant capacity.
The chemical analysis of D. indica, conducted by Sindhura & Eswaraiah[22], revealed significant differences between the ethanolic and chloroform fractions in the content of phenolic compounds and flavonoids, both of which contribute to the plant's antioxidant activity. A substantially higher content of phenolic compounds was observed in the ethanolic extract (71.54±3.58 mg GAE/g dry extract) compared to the chloroform fraction (35.29±4.36 mg GAE/g). Furthermore, the flavonoid content was also greater in the ethanolic fraction (64.51±1.94 mg RE/g dry extract) when compared to the chloroform fraction (28.51±0.52 mg RE/g).
D. triphylla contains noxious compounds that are sequestered by the larvae of the Hamadryas laodamia butterfly. Although these compounds remain chemically unspecified, they are presumed to include alkaloids (16), glycosides (25), terpenes, phenols (14), and fatty acids (15), all of which confer defensive properties upon the plant against herbivores and pathogens[23]. The larvae of H. laodamia exploit the plant's toxicity by retaining these compounds in their bodies and subsequently using them as a chemical defense against predators[24].
Histochemical studies conducted on the staminate flowers of D. alata indicate that the secretory glands and colleters produce lipidic substances, which are soluble in ether and comprise a mixture of two to six oxygenated triterpenes (4). These substances react positively with Sudan Black B, Nile Blue, and Nadi's reagent, thereby confirming their complex composition[25].
b) Biological activity
The study by Srinivasula & Maram[26] investigated the neuroprotective effects of the ethanolic (EDI) and chloroform (CDI) extract fractions of D. indica in rats subjected to a model of cerebral ischemia-reperfusion, a condition associated with ischemic stroke characterized by cerebral oxygen and glucose deprivation. The research aimed to assess the extracts' capacity to mitigate oxidative stress-induced damage, improve behavioral and biochemical parameters, and potentially serve as a natural compound-based therapeutic alternative. The results demonstrated that the ethanolic extract (EDI) exhibited superior efficacy compared to the chloroform extract (CDI), both in restoring neurological function and in improving antioxidant parameters, as evidenced by increased levels of antioxidant enzymes (SOD, CAT, GPx, GSH) and decreased lipid peroxidation.
The study conducted by Sindhura & Eswaraiah[27] investigated the thrombolytic, anti-inflammatory, and neuroprotective properties of D. indica Wight extract fractions, utilizing both in vitro and in vivo models. The results presented indicate that D. indica contains bioactive compounds with significant therapeutic potential, particularly for the treatment of diseases associated with thrombosis, inflammation, and cerebral ischemia, such as stroke.
Although studies specifically on the therapeutic effects of the Dalechampia genus are limited, several compounds identified within it exhibit potent and well-established biological activities. Terpenoids, for instance, constitute one of the most significant families of natural compounds in the medicinal field. Various members of this class have been used for centuries for therapeutic purposes, particularly for their antibacterial, anti-inflammatory, and antitumor activities. Diterpenes of the labdane type, for example, have demonstrated a broad spectrum of biological activities, including antimicrobial, cytotoxic, anti-inflammatory, antiviral, and antiprotozoal effects[28]. Carvone, found in the species under consideration, is a monoterpenoid ketone present in the essential oils of numerous aromatic and medicinal plants. Recent pharmacological studies have revealed that carvone possesses multiple therapeutic properties, encompassing antibacterial, antifungal, antiparasitic, antineuraminidase, antioxidant, anti-inflammatory, and anticancer activities[29].
The study of Sindhura & Eswaraiah[27] investigated the protective effects of EDI and CDI fractions against damage induced by cerebral ischemia and reperfusion, using the bilateral carotid artery occlusion (BCCAO) model in rats. The presented results indicated a significant reduction in cerebral infarct size, cerebral edema, and glutamate and acetylcholinesterase levels, suggesting that D. indica fractions possess neuroprotective activity.
The leaf extract was obtained via a cold extraction method and subsequently analyzed by Tsirinirindravo & Andrianarisoa[30], who reported a crude extract of D. clematidifolia characterized by an opaque brown color and a pH of 5.98. The research aimed to evaluate the natural antibiotic potential of this extract against various bacterial strains. Susceptibility tests demonstrated that the extract exhibited a broad spectrum of activity, proving particularly effective against species from the Enterobacteriaceae family (including Shigella boydii, Escherichia coli, Alkalescens dispar, Hafnia alvei, Enterobacter cloacae) and the Micrococcaceae (Micrococcus luteus, Staphylococcus aureus). Furthermore, the extract also showed efficacy against Rhodococcus sp., Listeria monocytogenes, L. utelshi, and L. ivanovii.
Triterpenes, also present in the genus Dalechampia, constitute a broad group of bioactive compounds implicated in the mechanisms of action and pharmacological effects of various medicinal plants traditionally used in folk medicine to treat immune-related diseases. These compounds have been described as anti-inflammatory, antiviral, antimicrobial, and antitumor agents, additionally possessing immunomodulatory properties. Many triterpenes contribute to the resolution of immunological diseases; however, their effects are not always clearly correlated, thereby reinforcing the imperative to further explore the activities of this class[31].
Sindhura & Eswaraiah[27] employed the human red blood cell (HRBC) membrane stabilization method to assess the capacity of the EDI and CDI fractions to protect against inflammatory damage. The fractions exhibited substantial protection, with EDI demonstrating 67.39 ± 2.11% stabilization compared to the positive control, diclofenac, which achieved 90.18 ± 1.42% protection. Although the anti-inflammatory effect of the D. indica fractions was lower than that of diclofenac, it remains relevant and suggests that the plant possesses therapeutic potential for inflammatory conditions.
Alkaloid compounds are also present in the Dalechampia genus. Plant alkaloids constitute one of the broadest groups of natural products, with diverse applications. In traditional medicine, for instance, plants containing protoberberine-type alkaloids are utilized in China for their analgesic, antiseptic, and sedative properties. Similarly, in Indian and Islamic folk medicine, these plants are frequently employed to treat bleeding disorders, eye diseases, and as uterine muscle relaxants. Moreover, various actions of alkaloids on the immune system have been reported, including the induction and inhibition of gene expression, anti-inflammatory effects, antiproliferative activity, inhibition of the complement system, and induction of apoptosis. Regarding the digestive system, significant actions such as anti-diarrheal properties, inhibition of electrolyte transport, and anti-ulcer activities have been observed[32].
Flavonoids exhibit diverse biochemical and antioxidant effects, which are associated with a range of diseases such as cancer, Alzheimer's disease, and atherosclerosis. These compounds are linked to a broad spectrum of beneficial health effects and are considered indispensable components in various pharmaceutical, medicinal, and cosmetic applications[33]. They are responsible for multiple biological effects, including anti-hepatotoxic, anti-inflammatory, and anti-ulcer properties. Furthermore, they inhibit various enzymes, such as aldose reductase and cyclooxygenase, and are recognized as potent antioxidants with the ability to scavenge free radicals. In addition to their antiallergic and antiviral actions, these compounds offer protection against cardiovascular mortality and have demonstrated the capacity to inhibit the growth of cancer cells in vitro, as well as reduce tumor development in experimental models[34].
In the antioxidant tests conducted by Sindhura & Eswaraiah[22], the ethanolic fraction of D. indica exhibited low IC50 values in the DPPH (135.04±3.99 µg/ml), hydrogen peroxide (153.80±9.61 µg/ml), and nitric oxide (100.41±0.96 µg/ml) assays, which reflects high antioxidant efficiency. The requirement of a smaller amount of extract to inhibit 50% of the free radicals confirms the effectiveness of the phenolic compounds and flavonoids present in the ethanolic extract (EDI). In contrast, the chloroform fraction demonstrated higher IC50 values, indicating reduced efficiency in neutralizing free radicals.
Sindhura & Eswaraiah[27] observed that the EDI (ethanolic extract) and CDI (dichloromethane extract) fractions of D. indica exhibited significant thrombolytic activity, with clot lysis percentages of 23.96 ± 1.67% and 18.64 ± 2.09%, respectively. Although these results are lower than those observed with streptokinase (72.86 ± 1.29%), a reference thrombolytic, the findings nonetheless suggest that the plant's fractions possess the potential to dissolve clots, particularly when considering the comparison with the negative control (water), which did not induce relevant lysis[27]. The observed thrombolytic effect may be attributed to the presence of compounds such as flavonoids or other antioxidants within the fractions, which are known to modulate clot formation and dissolution, as posited by[27].
Phenolic compounds are also constituents of the Dalechampia genus. In recent years, significant emphasis has been placed on the potential health benefits of dietary polyphenols of plant origin, which are recognized as potent antioxidants. Data from epidemiological investigations and meta-analyses indicate that prolonged intake of diets rich in plant polyphenols offers protection against cancer, cardiovascular disease, diabetes, osteoporosis, and neurological disorders[35]. Another compound present in the flowers of Dalechampia species is limonene. Pharmacological studies indicate that limonene possesses various therapeutic activities, including antibacterial, anticancer, analgesic, anti-inflammatory, and immunomodulatory effects. It demonstrates efficacy against multi-resistant bacterial infections and exhibits preventive action in cancer models by acting on cellular pathways that control the growth and resistance of tumor cells. Furthermore, its anti-inflammatory properties show potential for treating inflammatory conditions such as colitis and pneumonia[36].
Tannins are powerful enzyme-inhibiting agents due to their complexation with enzyme proteins[37]. They can interact with and precipitate proteins such as gelatin, which is believed to be responsible for the astringency observed in many plants[38]. In addition to precipitating proteins, tannins are also capable of interacting with carbohydrates, bacterial cell membranes, and metal ions[39].
One group of glycosides found in plants is known as saponins, characterized by their ability to form foam in aqueous solutions. The main activities attributed to saponins in the scientific literature include hemolytic, molluscicidal, anti-inflammatory, antifungal, antileishmanial, antimicrobial, antiparasitic, cytotoxic and antitumor, and antiviral activit[21].
Quinones belong to a group of compounds possessing two conjugated ketone groups, classified as dienones. Quinones encompass various types, such as those derived from benzene (benzoquinones), naphthalene (naphthoquinones), and anthracene (anthraquinones), all of which demonstrate a wide variety of biological activities, including antitumor, antifungal, antibacterial, and antichagasic effects, among others[28]. The volatile components myrcene and limonene, when present in essential oil, can exert an anxiolytic effect dependent on their concentration. This effect is related to the olfactory system, generating a response in the central nervous system[40].
Histamine is recognized as one of the key regulators of physiological homeostasis in the human body. It is released by cells such as mast cells, basophils, and platelets, exerting diverse functions across various physiological systems, including the respiratory, cardiovascular, gastrointestinal, and central nervous systems. In the cardiovascular system, histamine acts to dilate blood vessels, increasing vascular permeability and decreasing peripheral resistance, which consequently leads to a drop in blood pressure, according to Couto et al.[41].
Camphene, or 2,2-dimethyl-3-methylenebicyclo[2.2.1]heptane, is a monoterpene found in small quantities, typically appearing as a mixture of d(1R,4S and 1S,4S) isomers. The d(1R,4S) isomer predominates in natural emissions[42]. Camphene, along with camphor and other volatile compounds, are frequently found as active components in essential oils or natural extracts. Camphor, a ketone, possesses diverse industrial applications and can be utilized as an insect repellent, a solvent for paints and varnishes, an antiseptic, an expectorant, and even as incense. Aromatic compounds are generally composed of organic molecules containing a benzene ring in their structure, a cyclic and stable arrangement of six carbon atoms. In 1865, August Kekulé elucidated the stability of benzene, attributing it to the alternation of single and double bonds within its structure[43]. Aromatic compounds are extensively employed in the production of fine chemicals, valued for their specific properties, such as therapeutic and antioxidant effects, with applications in the pharmaceutical and agrochemical sectors[44].
Conclusions
The genus Dalechampia represents a promising source of bioactive chemical compounds with both therapeutic and ecological potential. This review study compiled relevant information on the chemical composition and biological activities of the species within this genus, highlighting the presence of substances such as diterpenes, triterpenes, flavonoids, and alkaloids, all known for their antimicrobial, anti-inflammatory, antioxidant, and neuroprotective properties.
Although Dalechampia possesses a considerable diversity of compounds with pharmacological potential, there remains a notable lack of in-depth studies comprehensively investigating their bioactive properties in an integrated manner. The literature reviewed indicates that existing research often focuses on taxonomic and morphological aspects, consequently relegating the possible medical applications of the identified compounds to a secondary role.
This work reinforces the imperative for further research exploring the ecological and pharmacological interactions within the Dalechampia genus. Future studies could significantly expand knowledge regarding the efficacy and safety of these compounds, thereby promoting responsible and sustainable medicinal use, alongside contributing to the conservation of these species. The recognition of the potential of the Dalechampia genus in pharmacological science and biodiversity conservation could represent a substantial advance in both traditional and modern medicine.
Acknowledgments
To the Federal University of Espírito Santo, to the Foundation for the Coordination of Improvement of Higher Education Personnel (CAPES), and to the National Council for Scientific and Technological Development (CNPq) and FAPES.
References
- 1 Wurdack, K. J.; Davis, C. C. Malpighiales phylogenetics: Gaining ground on one of the most recalcitrant clades in the angiosperm tree of life. Americ J Bot. 2009; 96(8): 1551-1570. [ https://doi.org/10.3732/ajb.0800207 ].
» https://doi.org/10.3732/ajb.0800207 - 2 Stevens PF. Angiosperm Phylogeny Website. 2001. [accessed on: Oct. 23, 2024]. Available at: [ http://www.mobot.org/MOBOT/research/APweb/ ].
» http://www.mobot.org/MOBOT/research/APweb/ - 3 Davis CC, Webb CO, Wurdack KJ, Jaramillo CA, Donoghue MJ. Explosive radiation of Malpighiales supports a mid-Cretaceous origin of modern tropical rain forests. The Amer Natural. 2005; 165(3): E36-E65. [ https://doi.org/10.1086/428296 ].
» https://doi.org/10.1086/428296 - 4 The Angiosperm Phylogeny Group . An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot J Linn Society. 2016; 181(1): 1-20. [ https://doi.org/10.1111/boj.12385 ].
» https://doi.org/10.1111/boj.12385 - 5 Flora of Brazil 2020, "Flora do Brasil 2020". [accessed on: 29 Oct 2024]. Available at: [ http://floradobrasil.jbrj.gov.br/reflora/listaBrasil/ConsultaPublicaUC/ ].
» http://floradobrasil.jbrj.gov.br/reflora/listaBrasil/ConsultaPublicaUC/ - 6 Jussieu AL . Euphorbiaceae. Genera Plantarum . p.384-392. 1789. [ https://www.biodiversitylibrary.org/item/7125 ].
» https://www.biodiversitylibrary.org/item/7125 - 7 Webster GL. Conspectus of a new classification of the Euphorbiaceae. Taxon. 1975; 24(5-6): 593-601. [ https://doi.org/10.2307/1220725 ].
» https://doi.org/10.2307/1220725 - 8 Webster GL. Classification of the Euphorbiaceae. Ann Missouri Bot Garden. 1994a; 81(1): p. 3-32. [ https://doi.org/10.2307/2399908 ].
» https://doi.org/10.2307/2399908 - 9 Webster GL. Synopsis of the Genera and Suprageneric Taxa of Euphorbiaceae. Ann Missouri Botan Garden. 1994b; (81): 33-144. [ https://doi.org/10.2307/2399909 ].
» https://doi.org/10.2307/2399909 - 10 Pereira-Silva RA, Athiê-Souza S, Armbruster WS, Sales MF. Typification and reestablishment of the Linnaean name Dalechampia colorata (Euphorbiaceae). Taxon. 2018; 67(1): 186-190. [ https://doi.org/10.12705/671.12 ].
» https://doi.org/10.12705/671.12 - 11 Botanico HUB . Dalechampia (Euphorbiaceae). 2024. [ https://doi.org/10.2307/2443104 ].
» https://doi.org/10.2307/2443104 - 12 Flora of North America, " Dalechampia ". [accessed on: 26 Oct 2024]. Available at: [ https://floranorthamerica.org/Dalechampia ]. Last edited on: July 29, 2020.
» https://floranorthamerica.org/Dalechampia - 13 Armbruster WS, Howard JJ, Clausen TP, et al. Do Biochemical Exaptations Link Evolution of Plant Defense and Pollination Systems? Historical Hypotheses and Experimental Tests with Dalechampia Vines. The Americ Natural. 1997; 149 (3): 461-484. [ https://doi.org/10.1086/286000 ].
» https://doi.org/10.1086/286000 - 14 Silva DF. O gênero Dalechampia L. (Euphorbiaceae - Acalyphoidea) na região sul do Brasil. 2018. Dissertação (Mestrado em Botânica) - Universidade Federal do Rio Grande do Sul, Porto Alegre. [acesso em: 25 jul. 2025]. Disponível em: [ https://www.iat.pr.gov.br/sites/agua-terra/arquivos_restritos/files/documento/2020-12/projeto_dilana_52_16.pdf ].
» https://www.iat.pr.gov.br/sites/agua-terra/arquivos_restritos/files/documento/2020-12/projeto_dilana_52_16.pdf - 15 Sazima M, Sazima I, Carvalho-Okano RM. Floral biology of Dalechampia stipulacea and its pollination by Euglossa melanotricha Rev Bras Biol. 1985; 45(1/2): 85-93. [ https://doi.org/10.5555/19860218760 ].
» https://doi.org/10.5555/19860218760 - 16 Araújo MRS, Lima MAS, Silveira ER. Triterpenes and steroids of Dalechampia pernambucensis Baill. 20073118340, English, Journal article, UK, 0305-1978, Oxford, Elsevier. Biochem Systemat Ecol. 2007; 35(5): 311-313. [ https://doi:10.1016/j.bse.2006.10.008 ].
» https://doi:10.1016/j.bse.2006.10.008 - 17 Armbruster WS, Lee J, Baldwin BG. Macroevolutionary patterns of defense and pollination in Dalechampia vines: adaptation, exaptation, and evolutionary novelty. Proceed Nation Acad Sci. 2009; 106(43): 18085-18090. [ https://doi.org/10.1073/pnas.0907051106 ].
» https://doi.org/10.1073/pnas.0907051106 - 18 Webster GL, Armbruster WS. An unusual new species of Dalechampia (Euphorbiaceae) from Surinam. System Bot. 1982; p.484-488. [ https://doi.org/10.2307/2418680 ].
» https://doi.org/10.2307/2418680 - 19 World Flora Online. Dalechampia dioscoreifolia Poepp. 2024. [accessed on: 29 Oct 2024]. Available at: [ http://www.worldfloraonline.org/taxon/wfo-0000937266 ].
» http://www.worldfloraonline.org/taxon/wfo-0000937266 - 20 Armbruster WS , Keller S , Matsuki M , Clausen TP . "Pollination of Dalechampia magnolifolia (Euphorbiaceae) by Male Euglossine Bees" (1989). An Paper. 104. [ https://doi.org/10.2307/2445136 ].
» https://doi.org/10.2307/2445136 - 21 Srivastava S, Panchani DT, Modi N. Phytochemical Analysis and Antioxidant Activity of Dalechampia scandens var. Cordofona (Hochst.ex A.Rich) Muell. Arg. Indian J Nat Sci. 2022. [ https://doi.org/10.13140/RG.2.2.22870.63044/1 ].
» https://doi.org/10.13140/RG.2.2.22870.63044/1 - 22 Sindhura S, Eswaraiah MC. Evaluation of in vitro thrombolytic, anti-inflammatory and stroke protective effects of Dalechampia indica extract fractions. Int Res J Pharm. 2017; 8(10): 196-202. [ https://doi.org/10.7897/2230-8407.0810207
» https://doi.org/10.7897/2230-8407.0810207 - 23 Armbruster WS, Herzig AL, Clausen TP. Pollination of two sympatric species of Dalechampia (Euphorbiaceae) in Suriname by male euglossine bees. Am J Bot. 1992; 79(12): 1374-1381. [ https://doi.org/10.1002/j.1537-2197.1992.tb13747.x ].
» https://doi.org/10.1002/j.1537-2197.1992.tb13747.x - 24 Henderson CL. Field guide to the wildlife of Costa Rica. Austin: University of Texas Press, 2002. p. 44. [ https://archive.org/details/fieldguidetowild0000hend ].
» https://archive.org/details/fieldguidetowild0000hend - 25 Martins FM , Cunha-Neto IL , Pereira TM . Floral morphology and anatomy of Dalechampia alata Klotzsch ex Baill. (Euphorbiaceae), with emphasis on secretory structures. Braz J Biol. 2016; 76: 233-244. [ https://doi.org/10.1590/1519-6984.19514 ].
» https://doi.org/10.1590/1519-6984.19514 - 26 Srinivasula S, Maram CE. Effect of Dalechampia indica Wight Extract Fractions on Behavioral and Biochemical Abnormalities following Ischemia-Reperfusion Insult in Rats. Pharmacog J. 2018; 10(1): 39-44. [ https://doi.org/10.5530/pj.2018.1.8 ].
» https://doi.org/10.5530/pj.2018.1.8 - 27 Sindhura S, Eswaraiah MC. Comparative evaluation of crude extract fractions of Taxillus heyneanus and Dalechampia indica for antioxidant activity and phenolic content. IOSR J Pharm. 2017; 7(5): 53-60. [ https://doi.org/10.9790/3013-0705015360 ].
» https://doi.org/10.9790/3013-0705015360 - 28 de las Heras B, Hortelano S. Molecular basis of the anti-inflammatory effects of terpenoids. Inflamm Allergy Drug Targets. 2009 Mar; 8(1): 28-39. [ https://doi.org/10.2174/187152809787582534 ]
» https://doi.org/10.2174/187152809787582534 - 29 Bouyahya A, Mechchate H, Benali T, Ghchime R, Charfi S, Balahbib A, et al. Health Benefits and Pharmacological Properties of Carvone. Biomolecules. 2021 Dec 1; 11(12): 1803. [ https://doi.org/10.3390/biom11121803 ].
» https://doi.org/10.3390/biom11121803 - 30 Tsirinirindravo L, Andrianarisoa B. Antibacterial activities of the extract from the leaves of Dalechampia clematidifolia (Euphorbiaceae). Inter J Biol Chem Sci. 2009; 3(5): [ https://doi.org/10.4314/ijbcs.v3i5.51098 ].
» https://doi.org/10.4314/ijbcs.v3i5.51098 - 31 Ríos JL. Effects of triterpenes on the immune system. J Ethnopharmacol. 2010; 128(1): 1-14. [ https://doi.org/0.1016/j.jep.2009.12.045 ].
» https://doi.org/0.1016/j.jep.2009.12.045 - 32 Bribi N. Pharmacological activity of alkaloids: a review. Asian J Bot. 2018; 1(1): 1-6. [ https://doi.org/10.63019/ajb.v1i2.467 ].
» https://doi.org/10.63019/ajb.v1i2.467 - 33 Karak, P. Biological activities of flavonoids: an overview. Int J Pharm Sci Res. 2019; 10(4): 1567-1574. [ https://doi.org/10.13040/IJPSR.0975-8232.10 (4).1567-74].
» https://doi.org/10.13040/IJPSR.0975-8232.10 (4).1567-74 - 34 Agrawal AD. Pharmacological Activities of Flavonoids: A Review. Scopus Indexed [Internet]. 2011 Aug. 31; 4(2): 1394-8. [ https://doi.org/10.37285/ijpsn.2011.4.2.3 ].
» https://doi.org/10.37285/ijpsn.2011.4.2.3 - 35 Rahman MM, Rahaman MS, Islam MR, et al. Role of phenolic compounds in human disease: current knowledge and future prospects. Molecules. 2021; 27(1): 233. [ https://doi.org/10.3390/molecules27010233 ].
» https://doi.org/10.3390/molecules27010233 - 36 Chen X, Ding Y, Guan H, et al. The Pharmacological Effects and Potential Applications of Limonene From Citrus Plants: A Review. Nat Product Communic. 2024; 19(5): 1-12. [ https://doi.org/10.1177/1934578X241254229 ].
» https://doi.org/10.1177/1934578X241254229 - 37 Naczk M, Amarowicz R, Pink D, Shahidi F. Condensed tannins in canola hulls. J Agric Food Chem. 1994; 42(10): 2196-2200. [ https://doi.org/10.1021/jf9908401 ].
» https://doi.org/10.1021/jf9908401 - 38 Strumeyer DH, Malin MJ. Condensed tannins in grain sorghum: isolation, fractionation, and characterization. J Agric Food Chem. 1975; 23(5): 909-914. [ https://doi.org/10.1021/jf60201a019 ].
» https://doi.org/10.1021/jf60201a019 - 39 Leinmüller E, Steingass H, Menke KH. Tannins in ruminant feedstuffs. Biannual Collection of Recent German Contributions Concerning Development through Animal Research. 1991; 33: 9-62. [ https://doi.org/10.5424/sjar/2004022-73 ].
» https://doi.org/10.5424/sjar/2004022-73 - 40 Leite MP , Fassin Jr J , Baziloni EMF , et al . Behavioral effects of essential oil of Citrus aurantium L. inhalation in rats. Rev Bras Farmacogn. 2008; 18: 661-666. [ https://doi.org/10.1590/S0102-695X2008000500003 ].
» https://doi.org/10.1590/S0102-695X2008000500003 - 41 Couto S, Martins C, Borrego LM. Histamine and its receptors: From physiology to pathology. Rev Port Imunoalerg. 2023; 31(1): 13-22. [ https://doi.org/10.32932/rpia.2023.03.099 ].
» https://doi.org/10.32932/rpia.2023.03.099 - 42 O'Neil MJ. (Ed.). The Merck index: an encyclopedia of chemicals, drugs, and biologicals. RSC Publishing, 2013. [ https://doi.org/10.1021/jm068049o ].
» https://doi.org/10.1021/jm068049o - 43 Rocke AJ. Kekulé's benzene theory and the appraisal of scientific theories. In: Scrutinizing science: Empirical studies of scientific change. Dordrecht: Springer Netherlands. 1988; 145-161. [ https://doi.org/10.12988/ams.2014.48621 ].
» https://doi.org/10.12988/ams.2014.48621 - 44 Alamgir ANM. Secondary metabolites: Secondary metabolic products consisting of C and H; C, H, and O; N, S, and P elements; and O/N heterocycles. In: Therapeutic use of medicinal plants and their extracts: volume 2: phytochemistry and bioactive compounds. Cham: Springer International Publishing, 2018. p.165-309. [ https://doi.org/10.1007/978-3-319-92387-1_3 ].
» https://doi.org/10.1007/978-3-319-92387-1_3
-
Funding Sources:
The authors declare no funding sources.
Publication Dates
- Publication in this collection
20 Apr 2026 - Date of issue
Apr 2026
History
- Received
02 Aug 2025 - Accepted
04 Mar 2026
 Review of the Chemical and Biological Properties of the Genus Dalechampia sp.
Review of the Chemical and Biological Properties of the Genus Dalechampia sp.
 Source: Authors' production (2025).
Source: Authors' production (2025).