Abstract
Fungal diseases are one of the most critical causes of postharvest losses in agricultural production, demanding safer and more sustainable control strategies. This study evaluated the chemical composition and in vitro antifungal activity of essential oils (EOs) from Lippia alba and Lippia turbinata against Botrytis cinerea, Monilinia fructicola, Rhizopus stolonifer, Colletotrichum nymphaeae, Fusarium hainanense, and Penicillium digitatum. The volatile method was applied using a concentration of 1000 ppm, with carbendazim as the chemical control. The EO of L. alba completely inhibited the growth of all fungi except P. digitatum (47.6 ± 1.4%), while L. turbinata showed 100% inhibition for all tested species, without significant differences compared to the fungicide (p>0.05). Gas chromatography–mass spectrometry analysis revealed β-linalool (26.72%) and trans-dihydrocarvone (16.29%) as major components in L. alba, and (−)-carvone (58.23%) and D-limonene (27.72%) in L. turbinata. The strong inhibitory activity and distinct chemical profiles suggest the potential of both species as sources of natural biofungicides for postharvest disease management.
Keywords Volatile compounds; antifungal activity; postharvest pathogens; native plants
Keywords Volatile compounds; antifungal activity; postharvest pathogens; native plants
Introduction
Over the past decades, fungi have been responsible for an increasing number of epidemics affecting humans, plants, and animals1. Globally, phytopathogenic fungi represents a major threat to agricultural productivity. Postharvest fungal diseases constitute one of the most critical challenges to global food security due to their impact on crop losses, food quality, and safety. Economic losses due to storage diseases can exceed those caused in the field. In developed countries, this loss can represent up to 25%; while in developing countries it is often higher, exceeding 50%[2–4].
In this regard, among the wide range of postharvest pathogens responsible for these losses, several species stand out for their high prevalence and destructive capacity. Botrytis cinerea is one of the most devastating species, affecting numerous crops worldwide and thriving under storage and transport conditions[5,6]. Similarly, Colletotrichum species represent a complex genus associated with anthracnose diseases that collectively account for more than 50% of global fruit and vegetable losses[7,8]. Fusarium species are also of major concern due to their cosmopolitan distribution, ability to cause severe plant diseases such as root rot and vascular wilt, and production of chemically stable mycotoxins, including fumonisins, zearalenone, and trichothecenes, that contaminate up to half of harvested crops annually[9,10]. Likewise, Rhizopus stolonifer is among the fastest-growing postharvest pathogens, capable of penetrating uninjured fruit cuticles and causing rapid tissue liquefaction under humid conditions[11–13]. Similarly, Penicillium digitatum, the causal agent of green mold in citrus fruits, spreads rapidly under humid, moderate conditions and has developed resistance to common fungicides such as imazalil and thiabendazole, making its control increasingly difficult[14,15]. Collectively, these pathogens represent a major challenge for the development of effective and sustainable postharvest disease management strategies. Finally, Monilinia fructicola, the causal agent of brown rot in stone and pome fruits, leads to significant pre- and postharvest losses and has shown a moderate risk of developing resistance to conventional fungicides[16].
Given the growing prevalence of resistant strains and the environmental concerns associated with synthetic fungicides, there is an urgent need for sustainable alternatives that align with the United Nations’ Sustainable Development Goals (SDG 2 and SDG 12), while meeting consumer and market demands for safer, low-residue produce[17–22]. Environmental impacts, including soil and water contamination, effects on non-target organisms, and disruption of ecological balance, have also raised serious concerns[23,24].
One of the most important plant defenses is the production of secondary metabolites that possess a wide range of biological activities. Natural plant products offer a set of structurally different antifungal agents that could represent an alternative to synthetic fungicides for the control of phytopathogenic fungi[3,25]. These compounds not only inhibit fungal growth but also enhance plant defense mechanisms, thereby contributing to sustainable postharvest disease management both in vitro and in vivo, and extending the overall quality and shelf life of fresh produce[26,27]. Within this framework, several plant-derived essential oils have shown remarkable antifungal potential. These compounds are selectively active against various fungal species, biodegradable, and potentially suitable for use as agrochemicals in integrated disease management programs[23,24]. Within this context, species of the genus Lippia have attracted particular attention due to their chemical diversity coupled with strong biological activity.
Lippia alba (Mill.) N.E.Br. ex Britton & P. Wilson and Lippia turbinata Griseb. (Verbenaceae) have emerged as promising botanical candidates for the postharvest control of phytopathogenic fungi. L. alba, commonly known as “salvia morada,” is widely distributed throughout tropical and subtropical regions of South America, including riparian forests in Argentina[28], and has a long-standing use in traditional medicine for gastrointestinal, respiratory, and pain-related disorders[29] (FIGURE 1 A). Its essential oil has demonstrated strong inhibitory activity against a wide range of fungal species, including aflatoxigenic Aspergillus flavus[29] and several plant pathogens such as Ustilago scitaminea, Cochliobolus falcatum, and Curvularia lunata[26], as well as Fusarium spp., Penicillium funiculosum, and Sclerotinia sclerotiorum[27]. Similarly, L. turbinata, known as “Poleo” and native to central and western Argentina[30,31], has shown antifungal potential; exposure of peanut seeds to its essential oil vapors effectively prevented contamination by Aspergillus species[29] (FIGURE 1 B).
This study aimed to evaluate the antifungal efficacy in vitro and chemical composition of L. alba and L. turbinata essential oils, with emphasis on their potential as biofungicides for postharvest disease management.
Material and Methods
Essential oil and plant material
The plants were collected from farms and roadsides in areas surrounding the Litoral region of Argentina between 2019 and 2020. Each plant sample was taxonomically identified, and a voucher specimen was deposited in the Herbarium “Arturo Ragonese” of the Facultad de Ciencias Agrarias, Universidad Nacional del Litoral (Herbarium SF), Kreder 2805, (3080HOF), Esperanza, Argentina: Lippia alba JFP13266 y Lippia turbinata JFP11609. Fresh leaves of each species were subjected to steam distillation using a Clevenger-type apparatus. The plant material was weighed to determine the essential oil yield per kilogram of processed fresh material. The obtained essential oils were stored in amber glass vials under refrigeration until further use.
Fungal strains
The phytopathogenic fungi Botrytis cinerea, Monilinia fructicola, Rhizopus stolonifer, Colletotrichum nymphaeae, Fusarium hainanense, and Penicillium digitatum were used in this study. B. cinerea was characterized and deposited in the Mycology Reference Center (CEREMIC) of the Facultad de Ciencias Bioquímicas y Farmacéuticas, Universidad Nacional de Rosario (FCByF–UNR), under the code CCC-100.39. M. fructicola was characterized and deposited in the Phytopathology Department of the San Pedro Experimental Station, Instituto Nacional de Tecnología Agropecuaria (INTA), Argentina, under the code INTA-SP345. R. stolonifer was characterized and deposited in the Microbiology Laboratory of the Facultad de Ingeniería Química, Universidad Nacional del Litoral (FIQ–UNL), Santa Fe, Argentina, under the code LMFIQ-317. C. nymphaeae and F. hainanense were characterized by the Phytopathology Chair of the Facultad de Ciencias Agrarias, Universidad Nacional del Litoral (FCA–UNL), and deposited in the Mycology Reference Center (CEREMIC) of FCByF–UNR under the codes CEREMIC 13-2019 and CEREMIC 01-2019, respectively. P. digitatum was characterized and deposited in CEREMIC, under the code CCC-102.
In vitro antifungal activity of essential oils using the volatile method
Petri dishes (60 mm in diameter) were filled with 20 mL of PDA medium. Once solidified, each plate was inoculated with a conidial suspension of 10⁴ CFU/mL placed into a well located at the center[32]. After the water from the inoculated conidial suspension had evaporated, 10 μL of the essential oils of Lippia alba and Lippia turbinata were deposited on the inner side of the plate lids, while sterile water was used as a growth control (T+). A commercial dose of carbendazim was used as a chemical control (T−). The amount of essential oil applied corresponded to a concentration of 1000 ppm. The Petri dishes were incubated upside down to allow the essential oil vapors, once evaporated, to come into contact with the culture medium where the fungus was growing (methodology adapted from Álvarez-Castellanos et al.[33]. When the mycelium of the control plates completely covered the surface of the medium (approximately 7 days), the mycelial diameter of each plate treated with essential oil was measured by scanning the plates (LA2400 scanner, WinRHIZO 2013 software; Regent Instruments Canada Inc., 2013) for subsequent analysis using ImageJ® software[34]. All assays were performed in triplicate, and the percentage of fungal growth inhibition was calculated according to the following equation: I% = 100 (C − M)/C, where I% represents the inhibition percentage, C is the average mycelial area of the three control plates, and M is the average mycelial area of the three treated plates. Confidence intervals (95%) for the mean mycelial growth inhibition of each treatment were calculated with a significance level of α = 0.05 using R software[35].
Identification of volatile constituents of essential oils
The essential oil was subjected to gas chromatography coupled with mass spectrometry (GC-MS) to identify its main components. The compounds were identified by comparing their mass spectra with those available in the NIST 2011 database. The sample was analyzed using a gas chromatograph (Agilent model 7890B) coupled to a mass spectrometer (Agilent model 5977) equipped with an HP-5MS UI capillary column (30 m × 0.25 mm, film thickness 0.25 μm). The operating conditions were as follows: injector column temperature 250°C; oven temperature initially set at 160°C and held for 3 min, then increased at 5°C min⁻1 up to 300°C; total run time: 31 min. Injection volume: 1 μL; split ratio: 1:20, according to the method described by Adams[36].
Results and Discussion
Yield of Essentials oils
Essential oils were obtained through steam distillation, which is recognized as the most widely used extraction method among several techniques such as enfleurage, expression, and fermentation[37–39]. In the present study, the extraction yield was 0.54% for Lippia alba and 0.56% for Lippia turbinata.
Previous studies have shown that the yield of L. alba varies seasonally, with higher values reported in summer (0.15-0.61%), autumn (0.47%), and spring (0.55%)[40]. In Argentina, authors have reported a range of 0.6-2% (mL/100 g of dry sample) in four chemotypes evaluated over two years[41].
In the case of L. turbinata, the yields obtained in this study are consistent with those previously reported in the literature[31]. Secondary metabolites responsible for biological activity are usually present in low concentrations in plant material, as observed in this work. Their extraction, purification, and characterization remain major challenges in the development of new pharmaceuticals or agrochemicals[42].
Antifungal in vitro activity
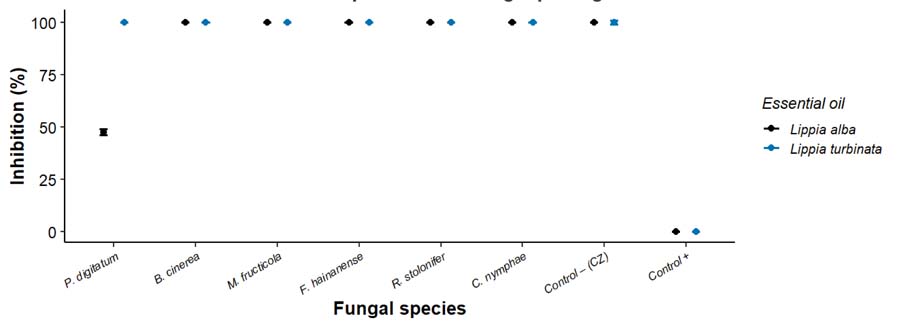
Complete inhibition (100%) was observed for Botrytis cinerea, Fusarium hainanense, Monilinia fructicola, Rhizopus stolonifer, and Colletotrichum nymphaeae by the essential oil (EO) of Lippia alba (FIGURE 2). In contrast, Penicillium digitatum exhibited partial sensitivity, with an average inhibition of 47.6 ± 1.4 %. Statistical analysis revealed that the inhibition of P. digitatum was significantly lower (p < 0.05) than that observed for the other tested fungi, indicating differential susceptibility among species.
: In vitro inhibitory effect of Lippia alba and Lippia turbinata essential oils against postharvest fungal pathogens. Points represent mean percentage inhibition, and error bars indicate 95% confidence intervals. Fungal species tested include Penicillium digitatum, Botrytis cinerea, Monilinia fructicola, Fusarium hainanense, Rhizopus stolonifer, and Colletotrichum nymphae. Negative (Control +) and chemical (Control – CZ) controls are included.
These results indicate a broad-spectrum antifungal activity, consistent with previous reports. Peixoto et al.[43] described similar minimum inhibitory concentrations, particularly against Lasiodiplodia theobromae, Fusarium pallidoroseum, and F. solani, achieving total inhibition at 0.2 mL/100 mL. The inhibitory effect of L. alba EO against Alternaria spp., as reported by Tomazoni et al.[44], further supports the broad anti-fungal potential of this species. In that study, full growth inhibition was achieved at relatively low doses, highlighting the ability of monoterpenes to disrupt fungal membrane permeability and ergosterol content. Similarly, Sabali et al.[45] reported complete inhibition of Aspergillus flavus at concentrations between 100 and 1000 ppm, demonstrating strong mycelial growth reduction for Aspergillus-related species. This is particularly relevant for food protection, as A. flavus is a well-known producer of aflatoxins. Therefore, L. alba EO could serve as a natural antifungal alternative to prevent post-harvest contamination. Finally, Arruda et al.[27] showed that L. alba EOs from the Brazilian Chaco, dominated by linalool, were highly effective against Sclerotinia sclerotiorum, achieving total growth inhibition.
On the other hand, complete inhibition (100%) was observed for all tested phytopathogenic fungi using the essential oil (EO) of L. turbinata, showing no statistically significant differences compared to the chemical control (p>0.05). These results indicate that the EO matched the efficacy of the synthetic fungicide under the tested conditions. The antifungal activity observed is consistent with previous reports on L. turbinata and related species. For instance, Passone and Etcheverry[29] demonstrated that volatile fractions from L. turbinata markedly inhibited Aspergillus section Flavi during peanut storage. Similarly, Leal et al.[46] confirmed the strong inhibitory capacity of L. turbinata EO across multiple fungal genera, supporting its broad-spectrum antifungal potential. Furthermore, ethanolic extracts of L. turbinata evaluated by Sayago et al.[47] exhibited fungicidal activity against Verticillium dahliae, reinforcing the potential of this species as a natural source of antifungal compounds. Likewise, Dellacassa et al.[48] reported variable levels of fungicidal activity of L. turbinata and L. integrifolia essential oils against Ascosphaera apis, an ascomycete fungus, confirming that differences in chemical profiles among Lippia species can influence their biological efficacy.
Chemical Composition
Essential oils (EOs), as the main components of the plant volatilome, are complex mixtures of volatile, lipophilic, and aromatic compounds whose qualitative and quantitative composition determines their biological activity[49].
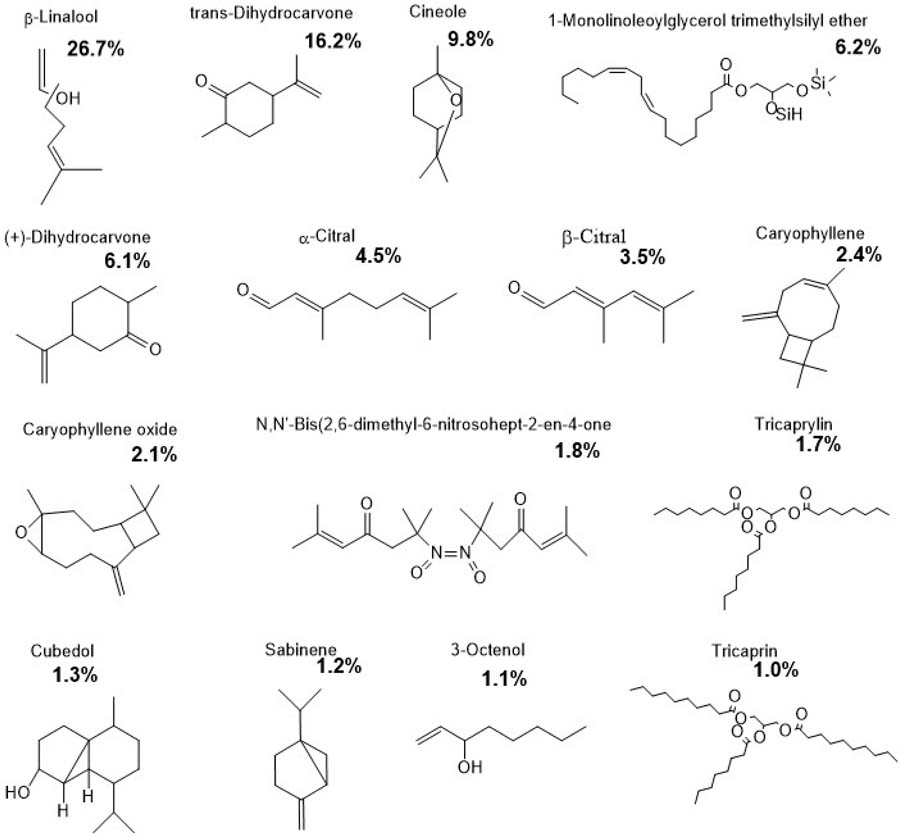
The essential oil of Lippia alba was mainly characterized by β-linalool (26.72%), trans-dihydrocarvone (16.29%), cineole (9.28%), dihydrocarvone (6.19%), α-citral (4.58%), β-citral (3.51%), caryophyllene (2.41%) and caryophyllene oxide (2.19%). Minor constituents, including sabinene (1.20%) and 1-octen-3-ol (1.14 %), accounted for a total of 86.2% of the oil composition (FIGURE 3). This chemical profile shows a predominance of oxygenated monoterpenes, particularly β-linalool and dihydrocarvone, consistent with the L. alba chemotype previously reported in Santa Fe, Argentina[41,50].
: Chemical composition of Lippia alba essential oil showing the major and minor constituents identified by GC–MS. The predominant compounds were β-linalool (26.7%), trans-dihydrocarvone (16.2%), and cineole (9.8%), followed by 1-monolinoleoylglycerol trimethylsilyl ether (6.2%), (+)-dihydrocarvone (6.1%), α-citral (4.5%), β-citral (3.5%), and caryophyllene (2.4%). Other components detected in lower proportions included caryophyllene oxide (2.1%), N,N′-bis(2,6-dimethyl-6-nitrosohept-2-en-4-one) (1.8%), tricaprylin (1.7%), cubedol (1.3%), sabinene (1.2%), 3-octenol (1.1%), and tricaprin (1.0%).
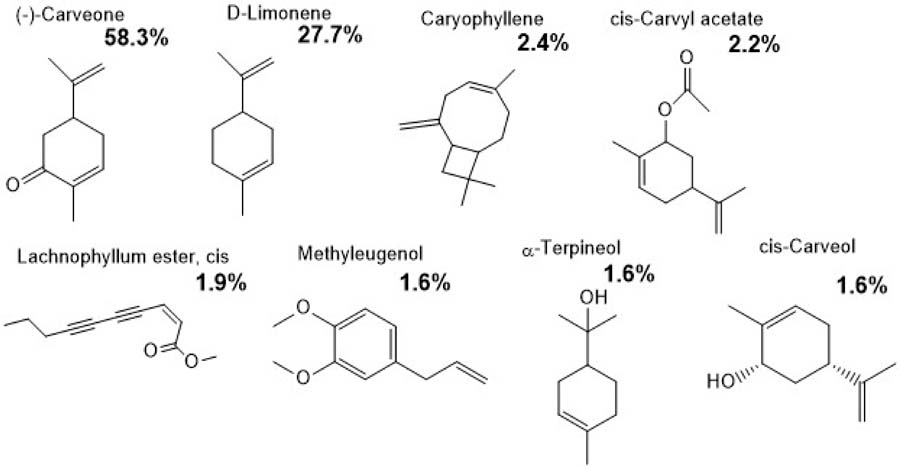
In contrast, the essential oil of Lippia turbinata exhibited a composition dominated by (-)-carvone (58.23%) and D-limonene (27.72%), followed by caryophyllene (2.48%), cis-carvyl acetate (2.25%), cis-lachnophyllum ester (1.90%), methyl eugenol (1.66%), α-terpineol (1.64%) and cis-carveol (1.61%), representing 97.49% of the total oil (FIGURE 4). This high percentage of identified compounds reflects the purity of the oil and agrees with the findings of Corzo et al.[31], who performed a meta-analysis of Argentine L. turbinata populations and reported consistent carvone–limonene chemotypes, suggesting strong genetic control of this profile.
: Chemical composition of Lippia turbinata essential oil showing the major and minor constituents identified by GC–MS. The predominant compounds were (–)-carvone (58.3%) and D-limonene (27.7%), followed by caryophyllene (2.4%) and cis-carvyl acetate (2.2%). Other components detected in lower proportions included lachnophyllum ester, cis (1.9%), methyleugenol (1.6%), α-terpineol (1.6%), and cis-carveol (1.6%).
The chemical comparison between the two Lippia species reveals clearly differentiated patterns: L. alba shows a predominance of β-linalool and cineole, while L. turbinata is rich in carvone and limonene. Such variability has been attributed to both genetic and environmental factors influencing EO composition[31,49]. In L. alba, β-linalool and cineole have been associated with antifungal and antimicrobial activities, supporting in vitro results against several fungal genera[25,26].
Carvone and limonene are the main monoterpenes responsible for their antimicrobial and repellent properties in L. turbinata. These compounds exhibit strong inhibitory activity against major phytopathogenic fungi such as Aspergillus, Fusarium, and Penicillium[46,51]. Moreover, the presence of minor constituents such as methyl eugenol and cis-carveol may contribute to synergistic effects that enhance the overall bioactivity of the oil. Girardi et al.[30] demonstrated that microencapsulation of L. turbinata EO preserved its antifungal efficacy over time, reinforcing the hypothesis that the combined action of major and minor terpenoids is crucial for the stability and persistence of its biological activity.
Conclusion
The results provide a scientific basis for the development of essential oils formulations from Lippia species, contributing to safer and more sustainable fungal control strategies in postharvest systems.
References
- 1 Hui ST, Gifford H, Rhodes J. Emerging Antifungal Resistance in Fungal Pathogens. Curr Clin Micro Rpt. 2024; 11(2): 43-50. Available from: [ https://doi.org/10.1007/s40588-024-00219-8 ].
» https://doi.org/10.1007/s40588-024-00219-8 - 2 Eckert JW, Ogawa JM. The chemical control of postharvest diseases: subtropical and tropical fruits. Annu Rev Phytopathol. 1985; 23(1): 421-54. Available from: [ https://assets.syngentaebiz.com/images/Eckert%201985.pdf ].
» https://assets.syngentaebiz.com/images/Eckert%201985.pdf - 3 Priyadarshi R, Routroy S, Garg GK. Analysis of post-harvest supply chain impediments for rural employability and waste reduction. IJSOM. 2022; 41(1/2): 163. Available from: [ https://doi.org/10.1504/IJSOM.2022.121693 ].
» https://doi.org/10.1504/IJSOM.2022.121693 - 4 Priyadarshi R, Routroy S, Garg GK. Postharvest supply chain losses: A state-of-the-art literature review and bibliometric analysis. JAMR. 2021; 18(3): 443-67. Available from: [ https://doi.org/10.1108/JAMR-03-2020-0040 ].
» https://doi.org/10.1108/JAMR-03-2020-0040 - 5 Dean R, Van KJAL, Pretorius ZA, Hammond-Kosack KE, Di Pietro A, Spanu PD, et al. The Top 10 fungal pathogens in molecular plant pathology. Mol Plant Pathol. 2012; 13(4): 414-30. Available from: [ https://doi.org/10.1111/j.1364-3703.2011.00783.x ].
» https://doi.org/10.1111/j.1364-3703.2011.00783.x - 6 Elad Y, Williamson B, Tudzynski P, Delen N, editors. Botrytis: Biology, Pathology and Control. Dordrecht: Springer Netherlands; 2007. Available from: [ https://doi.org/10.1007/978-1-4020-2626-3 ].
» https://doi.org/10.1007/978-1-4020-2626-3 - 7 Zakaria L. Diversity of Colletotrichum species associated with anthracnose disease in tropical fruit crops- A review. Agric. 2021; 11(4): 297. Available from: [ https://doi.org/10.3390/agriculture11040297 ].
» https://doi.org/10.3390/agriculture11040297 - 8 Bordoh PK , Ali A , Dickinson M , Siddiqui Y , Romanazzi G . A review on the management of postharvest anthracnose in dragon fruits caused by Colletotrichum spp. Crop Prot. 2020; 130: 105067. Available from: [ https://doi.org/10.1016/j.cropro.2019.105067 ].
» https://doi.org/10.1016/j.cropro.2019.105067 - 9 Stepien L , Lalak-Kanczugowska J , Witaszak N , Urbaniak M . Fusarium Secondary Metabolism Biosynthetic Pathways: So Close but So Far Away. In: Mérillon JM , Ramawat KG , editores . Co-Evolution of Secondary Metabolites. Reference Series in Phytochemistry Cham: Springer International Publishing. 2020; 211-247 . Available from: [ https://doi.org/10.1007/978-3-319-96397-6_28 ].
» https://doi.org/10.1007/978-3-319-96397-6_28 - 10 Thrane U . Fusarium. Encyclopedia of Food Microbiology: Second Edition. Elsevier; 2014. p. 76-81 . [ https://doi.org/10.1016/B978-0-12-384730-0.00141-5 ].
» https://doi.org/10.1016/B978-0-12-384730-0.00141-5 - 11 Snowdon AL. Post-Harvest Diseases and Disorders of Fruits and Vegetables: Volume 1: General Introduction and Fruits (1 st ed.). CRC Press; 1990. Available from: [ https://doi.org/10.1201/b18214 ].
» https://doi.org/10.1201/b18214 - 12 Bautista-Baños S, Bosquez-Molina E, Barrera-Necha LL. Rhizopus stolonifer (soft rot). In: Postharvest decay. Elsevier; 2014. p. 1-44. Available from: [ https://doi.org/10.1016/B978-0-12-411552-1.00001-6 ].
» https://doi.org/10.1016/B978-0-12-411552-1.00001-6 - 13 Liu Q, Chen Q, Liu H, Du Y, Jiao W, Sun F, et al. Rhizopus stolonifer and related control strategies in postharvest fruit: A review. Heliyon. 2024; 10(8). Available from: [ https://www.doi.10.1016/j.heliyon.2024.e29522 ].
» https://www.doi.10.1016/j.heliyon.2024.e29522 - 14 Wang Z, Sui Y, Li J, Tian X, Wang Q. Biological control of postharvest fungal decays in citrus: a review. Crit Rev Food Sci Nutr. 2022; 62(4): 861-70. Available from: [ https://doi.org/10.1080/10408398.2020.1829542 ].
» https://doi.org/10.1080/10408398.2020.1829542 - 15 Bhatta UK . Alternative management approaches of citrus diseases caused by Penicillium digitatum (green mold) and Penicillium italicum (blue mold). Front Plant Sci. 2022; 12: 833328. Available from: [ https://doi.org/10.3389/fpls.2021.833328 ].
» https://doi.org/10.3389/fpls.2021.833328 - 16 Martini C, Mari M. Monilinia fructicola, Monilinia laxa (Monilinia rot, brown rot). In: Postharvest decay. Elsevier; 2014, p. 233-65. Available from: [ https://doi.org/10.1016/B978-0-12-411552-1.00007-7 ].
» https://doi.org/10.1016/B978-0-12-411552-1.00007-7 - 17 Islam T, Danishuddin TNT, Matin MN, Barai HR, Haque MA. Resistance Mechanisms of Plant Pathogenic Fungi to Fungicide, Environmental Impacts of Fungicides, and Sustainable Solutions. Plants. 2024; 13(19): 2737. Available from: [ https://doi.org/10.3390/plants13192737 ].
» https://doi.org/10.3390/plants13192737 - 18 Pimentão AR , Cuco AP , Pascoal C , Cássio F , Castro BB . Current trends and mismatches on fungicide use and assessment of the ecological effects in freshwater ecosystems. Environ Pollut. 2024; 347: 123678. Available from: [ https://doi.org/10.1016/j.envpol.2024.123678 ].
» https://doi.org/10.1016/j.envpol.2024.123678 - 19 Thakur A, Thakur B, Kumar R. Post-harvest management of medicinal and aromatic plants: Current trends and recent advances. JEOBP. 2025; 28(1): 1-23. Available from: [ https://doi.org/10.1080/0972060X.2025.2461495 ].
» https://doi.org/10.1080/0972060X.2025.2461495 - 20 Ivanov M, Ciric A, Stojkovic D. Emerging Antifungal Targets and Strategies. Int J Mol Sci. 2022; 23(5): 2756. Available from: [ https://doi.org/10.3390/ijms23052756 ].
» https://doi.org/10.3390/ijms23052756 - 21 Singh HP, Batish DR, Kohli RK. Allelopathic Interactions and Allelochemicals: New Possibilities for Sustainable Weed Management. Crit Rev Plant Sci. 2003; 22(3-4): 239-311. [ https://doi.org/10.1080/713610858 ].
» https://doi.org/10.1080/713610858 - 22 Feliziani E, Romanazzi G. Preharvest application of synthetic fungicides and alternative treatments to control postharvest decay of fruit. Stewart Postharvest Rev. 2013; 9(3): 1-6. Available from: [ https://doi.org/10.2212/spr.2013.3.3 ].
» https://doi.org/10.2212/spr.2013.3.3 - 23 Sui Y , Liao Q , Leng J , Chen Z . Eco-friendly biocontrol strategies for management of postharvest fungal decays in kiwifruit: A review. Int J Food Microbiol. 2025; 432: 111106. Available from: [ https://doi.org/10.1016/j.ijfoodmicro.2025.111106 ].
» https://doi.org/10.1016/j.ijfoodmicro.2025.111106 - 24 Kalkan F. Management of Postharvest Diseases via Eco-Friendly Technologies: A Review of Recent Research. Hortic. 2025; 11(9): 1056. Available from: [ https://doi.org/10.3390/horticulturae11091056 ].
» https://doi.org/10.3390/horticulturae11091056 - 25 Shukla R, Kumar A, Singh P, Dubey NK. Efficacy of Lippia alba (Mill.) N.E. Brown essential oil and its monoterpene aldehyde constituents against fungi isolated from some edible legume seeds and aflatoxin B1 production. Int J Food Microbiol. 2009; 135(2): 165-70. Available from: [ https://doi.org/10.1016/j.ijfoodmicro.2009.08.002 ].
» https://doi.org/10.1016/j.ijfoodmicro.2009.08.002 - 26 Singh G, Rao G, Kapoor PS, Singh OP. Chemical constituents and antifungal activity of Lippia alba Mill leaf essential oil. JMAPS. 2000; 22: 701-703. Available from: [ https://www.cabidigitallibrary.org/doi/full/10.5555/20013071705 ].
» https://www.cabidigitallibrary.org/doi/full/10.5555/20013071705 - 27 Arruda RCO, Victorio CP, Boaretto AG, Carollo CA, Farias CS, Marchetti CR, et al. Essential oil composition, antifungal activity and leaf anatomy of Lippia alba (Verbenaceae) from Brazilian Chaco. JMPR. 2019; 13(4): 79-88. [ https://doi.org/10.5897/JMPR2018.6700 ].
» https://doi.org/10.5897/JMPR2018.6700 - 28 Crovetto RM. Plantas utilizadas en medicina en el NO de Corrientes. Fundación Miguel Lillo. Fundación Miguel Lillo; 1981 Available from: [ https://www.lillo.org.ar/editorial/index.php/publicaciones/catalog/book/209 ].
» https://www.lillo.org.ar/editorial/index.php/publicaciones/catalog/book/209 - 29 Passone MA , Etcheverry M . Antifungal impact of volatile fractions of Peumus boldus and Lippia turbinata on Aspergillus section Flavi and residual levels of these oils in irradiated peanut. Int J Food Microbiol. 2014; 168-169: 17-23. [ https://doi.org/10.1016/j.ijfoodmicro.2013.10.009 ].
» https://doi.org/10.1016/j.ijfoodmicro.2013.10.009 - 30 Girardi NS , García D , Passone MA , Nesci A , Etcheverry M . Microencapsulation of Lippia turbinata essential oil and its impact on peanut seed quality preservation. Int Biodeter Biodegrad. 2017; 116: 227-233. [ https://doi.org/10.1016/j.ibiod.2016.11.003 ].
» https://doi.org/10.1016/j.ibiod.2016.11.003 - 31 Corzo FL, Calvo FE, Lizarraga EF, Marcial GE, Mercado MI. Essential Oil Profiles of Lippia turbinata (Verbenaceae) from Argentina: Insights from a Systematic Review and Meta-Analysis. Chem Open. 14(11): 2500203. [ https://doi.org/10.1002/open.202500203 ].
» https://doi.org/10.1002/open.202500203 - 32 M51 | Method for Antifungal Disk Diffusion Susceptibility Testing of Nondermatophyte Filamentous Fungi. Available from: [ https://clsi.org/shop/standards/m51/ ].
» https://clsi.org/shop/standards/m51/ - 33 Alvarez-Castellanos PP, Bishop CD, Pascual-Villalobos MJ. Antifungal activity of the essential oil of flowerheads of garland chrysanthemum ( Chrysanthemum coronarium ) against agricultural pathogens. Phytochemistry. 2001; 57(1): 99-102. [ https://doi.org/10.1016/S0031-9422 (00)00461-1].
» https://doi.org/10.1016/S0031-9422 (00)00461-1 - 34 Schindelin J, Rueden CT, Hiner MC, Eliceiri KW. The ImageJ ecosystem: An open platform for biomedical image analysis. Mol Reprod Dev 2015; 82(7-8): 518-529, [ https://doi.org/10.1002/mrd.22489 ].
» https://doi.org/10.1002/mrd.22489 - 35 R Core Team. R Studio-Vienna, Austria: R Foundation for Statistical Computing; 2023. Available from: [ https://www.R-project.org/ ].
» https://www.R-project.org/ - 36 Adams C . Secondary and Higher Order Structural Characterization of Peptides and Proteins by Mass Spectrometry. Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology, 2007; Available from: [ https://urn.kb.se/resolve?urn=urn:nbn:se:uu:diva-7861 ] .
» https://urn.kb.se/resolve?urn=urn:nbn:se:uu:diva-7861 - 37 Cai J, Yan R, Shi J, Chen J, Long M, Wu W, et al. Antifungal and mycotoxin detoxification ability of essential oils: A review. Phytother Res. 2022; 36(1): 62-72. Available from: [ https://doi.org/10.1002/ptr.7281 ].
» https://doi.org/10.1002/ptr.7281 - 38 Rasul MG. Extraction, isolation and characterization of natural products from medicinal plants. Int J Basic Sci Appl Comput. 2018; 2(6): 1-6. Available from: [ https://www.researchgate.net/publication/330702256 ].
» https://www.researchgate.net/publication/330702256 - 39 Matrose NA , Obikeze K , Belay ZA , Caleb OJ . Impact of spatial variation and extraction solvents on bioactive compounds, secondary metabolites and antifungal efficacy of South African Impepho [ Helichrysum odoratissimum (L.) Sweet]. Food Biosci. 2021; 42: 101139. Available from: [ https://doi.org/10.1016/j.fbio.2021.101139 ].
» https://doi.org/10.1016/j.fbio.2021.101139 - 40 Castro DM, Ming LC, Marques MOM. Biomass production and chemical composition of Lippia alba (Mill.) N.E.Br. EX Britt & Wilson in leaves on different plant parts in differen seasons. Acta Hortic. 2002; (569): 111-5. Available from: [ https://doi.org/10.17660/ActaHortic.2002.569.18 ].
» https://doi.org/10.17660/ActaHortic.2002.569.18 - 41 Blanco M. Rendimiento de biomasa y aceite esencial de quimiotipos de Lippia alba (Mill.) N. E. Brown en respuesta a las prácticas agronómicas, y sus propiedades farmacológicas [Tesis]. Universidad Nacional de La Plata; 2014 [citado 11 de octubre de 2025]. Available from: [ http://sedici.unlp.edu.ar/handle/10915/43581 ].
» http://sedici.unlp.edu.ar/handle/10915/43581 - 42 Brusotti G , Cesari I , Dentamaro A , Caccialanza G , Massolini G . Isolation and characterization of bioactive compounds from plant resources: The role of analysis in the ethnopharmacological approach. J Pharm Biomed Anal. 2014; 87: 218-28 . Available from: [ https://doi.org/10.1016/j.jpba.2013.03.007 ].
» https://doi.org/10.1016/j.jpba.2013.03.007 - 43 Peixoto MG, Blank AF, Arrigoni-Blank MF, Gagliardi PR, Melo JO, Nizio DAC, et al. Activity of essential oils of Lippia alba chemotypes and their major monoterpenes against phytopathogenic fungi. Biosci J. 2018; 34(5): 1136-1146. Available from: [ http://www.seer.ufu.br/index.php/biosciencejournal/article/view/39385/24200 ].
» http://www.seer.ufu.br/index.php/biosciencejournal/article/view/39385/24200 - 44 Tomazoni EZ, Pansera MR, Pauletti GF, Moura S, Ribeiro RTS, Schwambach J. In vitro antifungal activity of four chemotypes of Lippia alba (Verbenaceae) essential oils against Alternaria solani (Pleosporeaceae) isolates. An Acad Bras Ciênc. 2016; 88(2): 999-1010. Available from: [ https://doi.org/10.1590/0001-3765201620150019 ].
» https://doi.org/10.1590/0001-3765201620150019 - 45 Sabaly S, Tine Y, Diallo A, Faye A, Cisse M, Ndiaye A, et al. Antifungal Activity of Cyperus articulatus, Cyperus rotundus and Lippia alba Essential Oils against Aspergillus flavus Isolated from Peanut Seeds. J Fungi. 2024; 10(8): 591. Available from: [ https://doi.org/10.3390/jof10080591 ].
» https://doi.org/10.3390/jof10080591 - 46 Leal LE, Alarcon A, Ortega Baes FP, Cayo F, Alarcón R. Effects of essential oils from two Lippia species on growth of phytopathogenic fungi. 2019. Available from: [ https://notablesdelaciencia.conicet.gov.ar/handle/11336/127674 ].
» https://notablesdelaciencia.conicet.gov.ar/handle/11336/127674 - 47 Sayago M, Ormeño I, Ajmat MT, Barbieri N. Essential oils and extracts from Argentinian northwest plants as potential biofungicides for olive and grapevine pathogens: in vitro studies. Rev Fac Ciencias Agrar UNCuyo. 2025; Ahead of Print. Available from: [ https://revistas.uncu.edu.ar/ojs3/index.php/RFCA/article/view/7813 ].
» https://revistas.uncu.edu.ar/ojs3/index.php/RFCA/article/view/7813 - 48 Dellacasa AD, Bailac PN, Ponzi MI, Ruffinengo SR, Eguaras MJ. In vitro Activity of Essential Oils from San Luis-Argentina Against Ascosphaera apis. J Essent Oil Res. 2003; 15(4): 282-5. Available from: [ https://doi.org/10.1080/10412905.2003.9712143 ].
» https://doi.org/10.1080/10412905.2003.9712143 - 49 Almeida MC, Pina ES, Hernandes C, Zingaretti SM, Taleb-Contini SH, Salimena FRG, et al. Genetic diversity and chemical variability of Lippia spp. (Verbenaceae). BMC Res Notes. 2018; 11(1): 725. Available from: [ https://doi.org/10.1186/s13104-018-3839-y ].
» https://doi.org/10.1186/s13104-018-3839-y - 50 Blanco MA, Colareda GA, van Baren C, Bandoni AL, Ringuelet J, Consolini AE. Antispasmodic effects and composition of the essential oils from two South American chemotypes of Lippia alba J Ethnopharm. 2013; 149(3): 803-9. Available from: [ https://doi.org/10.1016/j.jep.2013.08.007 ].
» https://doi.org/10.1016/j.jep.2013.08.007 - 51 Passone MA , Girardi NS , Ferrand CA , Etcheverry M . In vitro evaluation of five essential oils as botanical fungitoxicants for the protection of stored peanuts from Aspergillus flavus and A. parasiticus contamination. Int Biodeter Biodegrad. 2012; 70: 82-8 . Available from: [ https://doi.org/10.1016/j.ibiod.2011.11.017 ].
» https://doi.org/10.1016/j.ibiod.2011.11.017
-
Funding:
Universidad Nacional del Litoral (CAI+D, 85420240100114LI) Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, PICT-2020SERIEA-02504).
Publication Dates
- Publication in this collection
18 Mar 2026 - Date of issue
Mar 2026
History
- Received
12 Dec 2025 - Accepted
06 Mar 2026
 Chemical composition and in vitro antifungal activity of essential oils from Lippia alba and Lippia turbinata
Chemical composition and in vitro antifungal activity of essential oils from Lippia alba and Lippia turbinata